With molecular diagnostics to biomarker-based personalised therapy
Diagnosing suitable biomarkers is a prerequisite for tailoring personalised therapies to patient heterogeneity. Genetic tests and genome sequencing play a key role in these diagnoses. Up until now, personalised therapy has achieved the greatest success in the field of oncology. However, personalised treatments are also gaining in importance for treating other diseases. In Baden-Württemberg, the expansion and further development of personalised treatments to other indications is mainly the realm of large interdisciplinary institutions such as the Heidelberg Centre for Personalised Oncology and the Centre for Personalised Medicine in Tübingen.
Personalised medicine is usually defined as a search for therapies that are tailored to the requirements of individual patients that involves finding the drugs that are best suited to treat a given patient’s particular condition. One would think that this has always been every good doctor’s goal. However, the paradigm shift brought about by personalised medicine, which has been coming for a good ten years, can only be achieved through a methodological approach. In 2016, immunologist Professor Hans-Georg Rammensee from Tübingen explained personalised medicine as follows: “One looks at a patient, analyses his or her biomarkers and then selects a drug from an existing arsenal of medicines that optimally matches a given patient.”
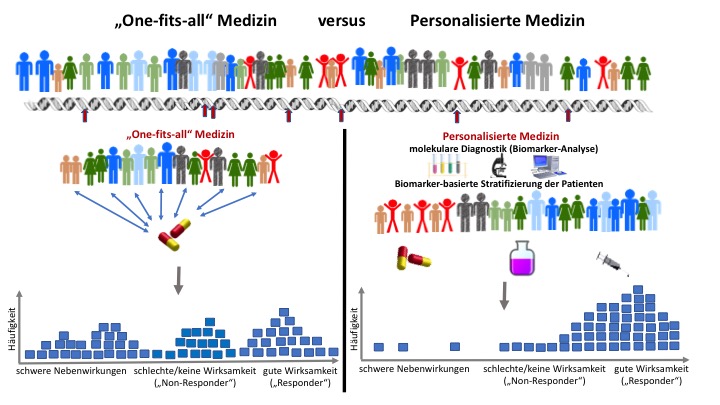 "One-size-fits-all" medicine versus "personalised medicine".
© EJ, modified from a figure provided by the Helmholtz Centre for Infection Research
"One-size-fits-all" medicine versus "personalised medicine".
© EJ, modified from a figure provided by the Helmholtz Centre for Infection Research
Personalised, individualised or stratified medicine
Rammensee explicitly referred to "stratified medicine", a term that has increasingly been used in recent years to replace "personalised medicine". The use of the term personalised has been questioned, because personalised medicine is not about individual characteristics such as self-knowledge or reflection, but about an individual’s molecular and biological makeup. The term “individualised medicine” is often used as a synonym for “personalised medicine” but is also a term that has been questioned. This is because rather than referring to a distinct individual, it usually designates the grouping of a patient into a subpopulation with a specific disease “subtype” that differs from other diagnosed subtypes through defined characteristics (“markers”). This is called stratification. However, this article is not about semantic differences and will therefore continue to use the term personalised medicine, which is still the most common of the three.
Companion diagnostics
Genome sequencing is a basic technology in personalised medicine; the photo shows the sequencing centre at the DKFZ.
© DKFZ
Diagnosing biomarkers is of crucial importance for this new approach to patient care. Biomarkers are used to determine a patient’s specific clinical picture and to select the most suitable drug to treat a patient’s disease. Accurate disease diagnosis is a prerequisite for personalised treatment. The development of an appropriate diagnostic test goes often hand in hand with or even precedes the development of a highly specific drug. This is called “companion diagnostics”, and is used to determine the effect of a therapeutic drug in a specific person.
For example, before the therapeutic antibody trastuzumab is used for treating metastatic breast cancer or stomach cancer, it is imperative to find out whether a particular patient’s tumour overexpresses a particular cell surface protein called epidermal growth factor receptor HER2, which this antibody targets. Similarly, the personalised treatment of colon and lung cancer requires knowledge and hence tests to find out whether the patient in question has mutations in the KRAS gene. Metastatic melanoma often contains a specific mutation in an oncogene called BRAF-V600, which can be identified in a test that detects the presence of a BRAF-V600 mutation and the cancer is then treated with targeted medicines.
There are still relatively few validated, clinically tested companion diagnostic tests. However, as the number of personalised therapies grows, the number of clinically tested companion diagnostic tests will undoubtedly rise considerably over the next few years. The Interreg project Codex4SMEs, which is being funded by the European Union from 2017 to 2020 with funds totalling 1.9 million euros and coordinated by BioRegio STERN Management GmbH in Stuttgart, promotes the development of companion diagnostics by small and medium-sized enterprises in seven countries in North-West Europe.
Genome sequencing of every cancer
Currently standing at around fifty, the number of drugs approved for personalised medicine applications in Germany is still rather low. But major progress can be expected. This expectation is based on the fact that out of all drug candidates currently in drug development pipelines at pharmaceutical companies, the proportion of personalised medicine drugs already accounts for 42 percent. If figures for preclinical phases of drug development are included, the proportion of personalised medicine drugs reaches 73 percent. Most of these drugs and drug candidates are in the oncology field, where personalised medicine has achieved the greatest success to date. As far as cancer is concerned, the inadequacies of the “one-size-fits-all” concept (the same therapy for all patients with – apparently – the same disease) become particularly obvious from the large number of drug intolerances, severe adverse drug effects and resistance to drugs that are usually used for the treatment of cancers.
The sequencing of individual tumour genomes brought to light an enormous genetic heterogeneity of tumours, even of similar ones. In many cases, thousands of different mutations occur. The analysis of the genomic changes in a tumour is now one of the crucial foundations of personalised oncology. The National Center for Tumour Diseases (NCT) in Heidelberg has the stated goal – which is not far from being reached – of determining the tumour genome of all of its tumour patients (about 3,500 per year) using a method known as whole genome sequencing.
The sequencers and the enormous computing power and data processing tools required to do this are provided by the German Cancer Research Center (DKFZ) in Heidelberg within the framework of the Heidelberg Center for Personalised Oncology (HIPO). The data volumes that accumulate on a daily basis (about 10 terabytes per day) are similar to the huge volumes generated by the social networking service Twitter. Such analyses also allow the identification of so-called driver mutations that are responsible for tumour development and growth. These driver mutations have the potential to be used as targets for new therapies or as predictive biomarkers (so-called risk indicators).
The sequencers and the enormous computing power and data processing tools required to do this are provided by the German Cancer Research Center (DKFZ) in Heidelberg within the framework of the Heidelberg Center for Personalised Oncology (HIPO). The data volumes that accumulate on a daily basis (about 10 terabytes per day) are similar to the huge volumes generated by the social networking service Twitter. Such analyses also allow the identification of so-called driver mutations that are responsible for tumour development and growth. These driver mutations have the potential to be used as targets for new therapies or as predictive biomarkers (so-called risk indicators).
The DKFZ-HIPO is based on three pillars:
- Genome Analysis programme led by Professor Peter Lichter
- Bioinformatics, Systems Biology and Synthetic Biology programme led by Professor Roland Eils
- Precision Oncology programme of the NCT, led by Professor Christof von Kalle.
DKFZ-HIPO integrates the clinical developments in new personalised drug candidates beyond the centre, thus maintaining ongoing collaborations with the pharmaceutical industry.
HIPO has become a pioneer in personalised oncology in Europe.
The Baden-Württemberg Cancer Registry and the Centre for Personalised Medicine (ZPM)
So that patients who live outside the catchment area of large cities and university hospitals can benefit from the new treatment concepts, relevant information must filter right through to general practitioners in rural areas. This is what the Baden-Württemberg Cancer Registry sets out to achieve. Every new cancer in Baden-Württemberg has to be reported to this registry, including detailed information on therapy and disease progression. The Head of the Clinical State Registry, Dr. Ing. Johannes Englert, has suggested that the cancer registry could be linked to genetic information and other personalised medicine data about a tumour in order to provide optimal treatment1; however, the combination of such data is unlikely in the near future due to data privacy and medical and bioethical concerns, similar to those that have been raised against the introduction of electronic health cards.
In addition to genomics, other "omics" technologies such as transcriptomics, epigenomics, proteomics and metabolomics are important for the individual analysis of the clinical picture and diagnostics that accompany therapy. These high-throughput procedures, together with modern imaging, are a priority at the Centre for Personalised Medicine (ZPM) in Tübingen, where 23 institutes and clinics have joined forces within the German Excellence Initiative with the aim of further developing personalised medicine approaches not just in the field of oncology2. The natural and information sciences are also involved in these developments. The chairman and spokesman of the ZPM is Professor Nisar Malek, medical director of the Department of Medicine I at Tübingen University Hospital. The huge data volumes arising from high-throughput technologies are processed by the Centre for Quantitative Biology (QBiC) at the University of Tübingen, which is led by Professor Oliver Kohlbacher.
Personalised therapy beyond the field of oncology
Active pharmaceutical ingredients that are used in the field of personalised medicine are not limited to cancer. One example is the treatment of HIV-infected persons with the reverse transcriptase inhibitor abacavir. Before HIV patients are given the drug, they need to undergo genetic testing to find out whether they are carrying a specific histocompatibility antigen (HLA-B*5701). The test is mandatory as carriers of this antigen have a life-threatening hypersensitivity to the drug. Genetic tests are also increasingly being used for treating hepatitis C infections. However, in this case the tests target the pathogen rather than the patient, as Professor Michael Manns, founding director of the Centre for Individualised Infection Medicine in Hannover, explains3: “An appropriate, effective therapy is selected depending on the subtype of the pathogen that has caused the disease. So the patient can be treated in a targeted way.”
As far as complex metabolic diseases that are associated with elevated cholesterol and fat levels are concerned, genetic tests can be used to stratify the patient population in order to select the most effective treatment strategy. Such applications are only just starting to be developed. Proteins and metabolic products that are indicative of pathological processes in the body can also be used as biomarkers and can be reliably measured at the ZPM in Tübingen using proteomics and metabolomics approaches. ZPM director Malek explicitly mentions heart failure, for which “drugs or drug combinations that have the highest chance of treatment success in a given patient can be selected from the wealth of theoretically applicable drugs using simulations based on omics data in conjunction with imaging methods”.
The new therapeutic approaches are also included in the funding programme "Digitisation in Medicine and Nursing", which was launched by the Baden-Württemberg Ministry of Social Affairs and Integration as part of the state's digitisation strategy called "digital@bw"4. The "Personalised Medicine Portal BW" was set up to accompany the further development of personalised medicine and its integration into standard treatment and care at the University Hospitals of Tübingen, Heidelberg, Ulm and Freiburg. It is a key project in the digital@bw strategy and is headed up by Professor Malek in Tübingen. The "bwHealthCloud" project led by Professor Kohlbacher at the QBiC in Tübingen is closely related to the same strategy. It aims to pool the clinical and molecular data on patient treatment from the four university hospitals working with personalised medicine, with a data management structure accessible to all doctors at these hospitals so that they can be used for therapy decisions and for following up similar cases in the future.