Researchers successfully rejuvenate the immune system in animal models
It is well known that stem cells age. Even the human immune system loses its power with age. Since all immune cells are derived from blood-forming stem cells, it is quite natural to associate the weakening of the immune system (immune senescence) with the ageing of blood-forming stem cells. Stem cell researchers and immunologists from the University of Ulm have now demonstrated the important role that blood-forming cells play in the ageing of the immune system in a new mouse model, thus disputing standard thinking.1
 Stem cell researchers and immunologists from Ulm have demonstrated that blood-forming stem cells have a major influence on the immune system. Photo (from left to right): Prof. Hartmut Geiger, Dr. Hanna Leins (Institute for Molecular Medicine), Prof. Reinhold Schirmbeck and Jun.-Prof. Medhanie Mulaw. © Andreas Brown
Stem cell researchers and immunologists from Ulm have demonstrated that blood-forming stem cells have a major influence on the immune system. Photo (from left to right): Prof. Hartmut Geiger, Dr. Hanna Leins (Institute for Molecular Medicine), Prof. Reinhold Schirmbeck and Jun.-Prof. Medhanie Mulaw. © Andreas BrownResearchers’ success in rejuvenating the defence system via stem cells could be of major clinical interest in the medium term. In earlier work, Prof. Dr. Hartmut Geiger, a stem cell expert, used the pharmacological substance CASIN (Cdc42 activity-specific inhibitor) to rejuvenate haematopoietic, i.e. blood-forming stem cells2. The latest test results eventually make it feasible to use this rejuvenating cure to reverse immune system ageing and improve influenza vaccination outcomes in elderly people.3 Professor Hartmut Geiger and Dr. Hanna Leins, first author of the paper published in the renowned science journal Blood, emphasise that there is still a long way to go before this knowledge can be applied.
Standard thinking refuted
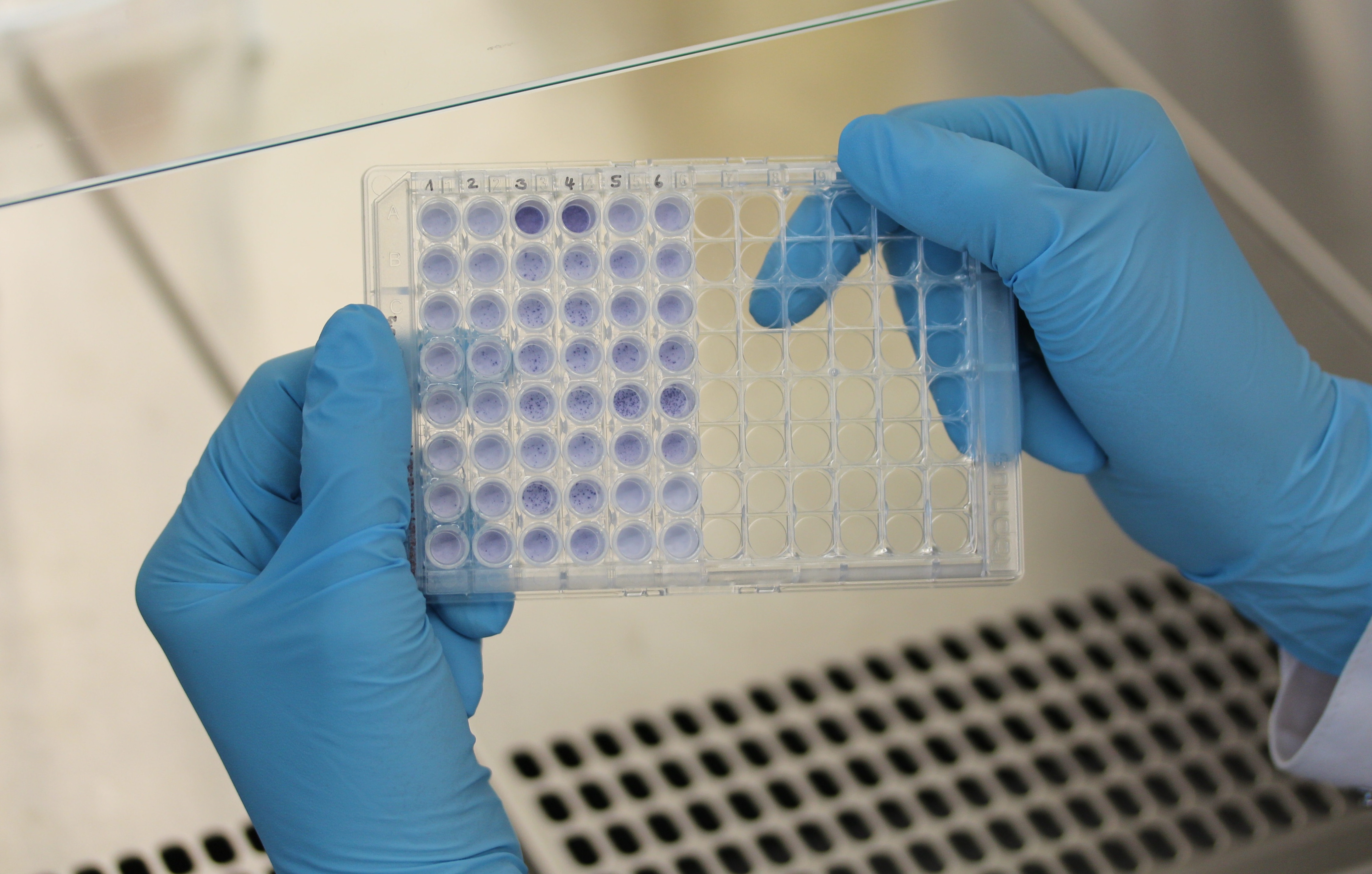 The researchers used an enzyme-linked immunospot assay to measure the vaccine response. The number of spots (blue dots) shows the strength of the immune response. © Photo: Andreas Brown
The researchers used an enzyme-linked immunospot assay to measure the vaccine response. The number of spots (blue dots) shows the strength of the immune response. © Photo: Andreas BrownIs there a causal connection between stem cell ageing and the ageing of immune cells, which originate from the very same stem cells? The thymus, which is responsible for the maturation and differentiation of T cells, shrinks after puberty and was long thought to be the major reason why the immune system weakens as we get older. The stem cell researchers from Ulm thought differently and started focusing on blood-forming stem cells instead. Not only because all efforts to activate the thymus had previously been unsuccessful, but also because most immune cells that respond to infection are very short-lived. They only live for a few weeks and have to be reproduced, something that is not compatible with the standard belief that T and B cells age too, thus contributing to the ageing of the immune system.
Together with immunologist Prof. Dr. Reinhold Schirmbeck from the Department of Internal Medicine I at Ulm University Hospital, Geiger and Leins looked for a system to verify their assumption. The group of immunologists led by Schirmbeck developed a mouse model of bone marrow transplantation based on the following idea: immune-deficient transgenic mice implanted with young or old stem cells develop a new immune system. This can be characterised phenotypically and functionally by immunising the animals and measuring their immune responses.
For this purpose, bone marrow stem cells were isolated from younger and older mice. Some of the older cells then underwent the researchers’ rejuvenation treatment. In a second step, the young, old and rejuvenated blood-forming stem cells were transplanted into transgenic mice without an immune system. The competence of the immune system was assessed after twelve weeks, amongst other things by looking at the mice’s vaccination response.
Important role played by blood-forming stem cells in the ageing of the body's immune defence
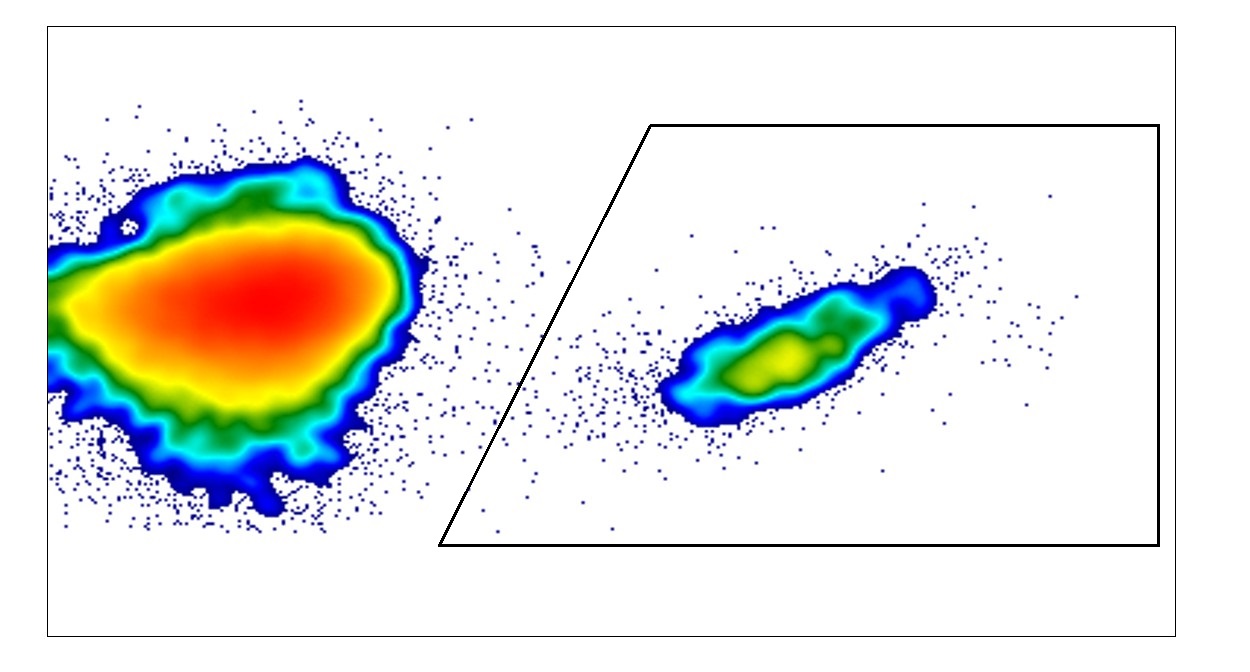 Flow cytometry analysis of the vaccination reaction: the immune system of young, old and rejuvenated stem cells generates many immune cells (framed in black) in response to a vaccine. The figure shows a density plot of the flow cytometry analysis; red indicates high cell density, and blue, low cell density. © Hanna Leins
Flow cytometry analysis of the vaccination reaction: the immune system of young, old and rejuvenated stem cells generates many immune cells (framed in black) in response to a vaccine. The figure shows a density plot of the flow cytometry analysis; red indicates high cell density, and blue, low cell density. © Hanna LeinsThe results proved the researchers right: animals transplanted with young stem cells developed a phenotypically and functionally young immune system similar to that of a young mouse. Animals transplanted with old stem cells ended up with a very similar immune system to that of old mice, i.e. they showed a much weaker response to vaccination. For immunologist Reinhold Schirmbeck the results prove “the important role of blood-forming stem cells in the ageing of the immune system.” Schirmbeck concludes that when stem cells age, the immune system is no longer able to sufficiently regenerate and the organism becomes more susceptible to infections.
The new model can be used to effectively create an immune system in a mouse in which only the immune system is allowed to age. The rejuvenation of the stem cells enables the researchers to find out whether the immune system’s clock can be turned back. Following the ex vivo experiments, the first in vivo experiments have already taken place. But Geiger explains that carrying out all the required in vivo experiments will take years. The researchers hope that the new model will provide them with new insights into what physiological changes an ageing immune system undergoes in comparison to a young one: does an 'old' immune system contribute to senescent cells (with cell cycle arrest) no longer being cleared so well from the tissue? Is there a connection between an old immune system and heart disease? What role does the ageing immune system play in neurodegenerative processes?
The researchers from Ulm are not ruling out the possibility that other factors might contribute to the ageing of the immune system. In the paper published in the scientific journal Blood, the researchers only used young animals. Similar experiments will now be carried out with old animals to find out what role the stem cell environment plays. It is known that the niche in the bone marrow where the stem cells live also ages.
Accurate presentation of the ageing of the immune system
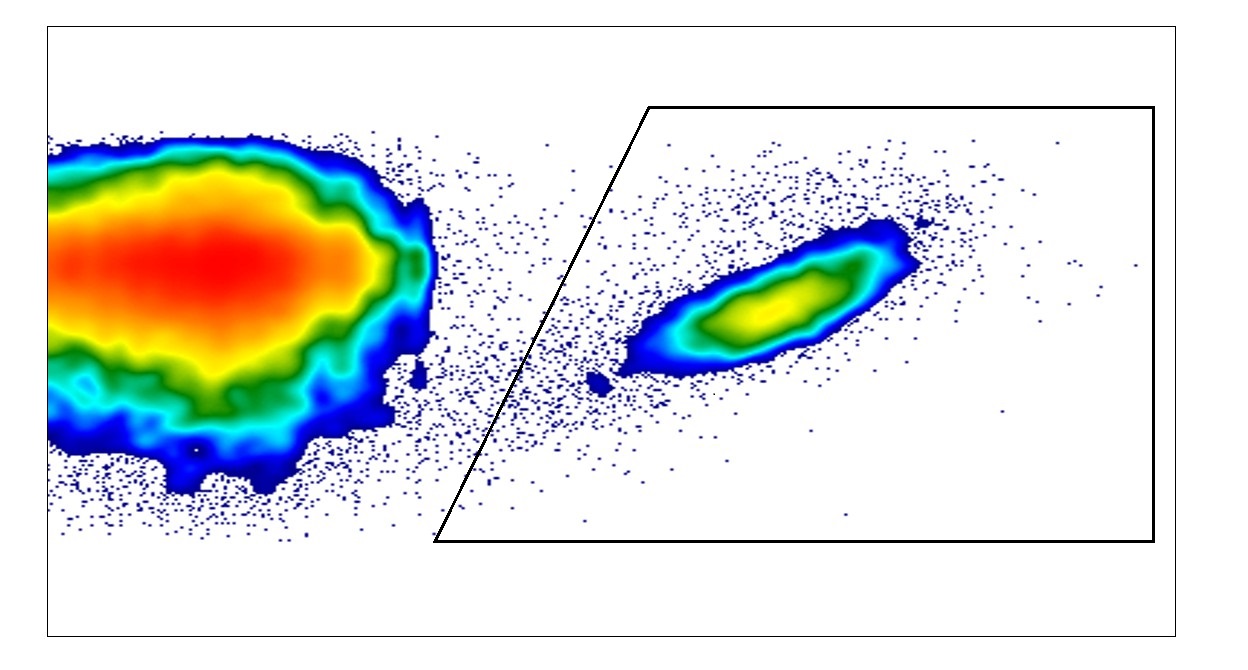 Flow cytometry analysis of the immunisation reaction: the immune system of young, old and rejuvenated stem cells generates many immune cells (framed in black) in response to a vaccine. The figure shows a density plot of the flow cytometry analysis; red indicates high cell density, and blue, low cell density. © Hanna Leins
Flow cytometry analysis of the immunisation reaction: the immune system of young, old and rejuvenated stem cells generates many immune cells (framed in black) in response to a vaccine. The figure shows a density plot of the flow cytometry analysis; red indicates high cell density, and blue, low cell density. © Hanna LeinsThe stem cell researchers’ major goal is to accurately represent the ageing of the immune system in the relatively controlled mouse environment. The researchers are seeking answers to two central questions: can the immune system in old mice be rejuvenated with a substance called CASIN when the mouse’s performance capacity is decreasing? And can the rejuvenated immune system clear away senescent cells that are thought to be responsible for us getting older and the development of age-associated diseases? Geiger believes that if this could be demonstrated this would be perfect. The pancreas, lungs, liver or skin would be suitable organs for such experiments. Prof. Dr. Karin Scharfetter-Kochanek, a dermatologist from Ulm University Hospital, is concentrating on experiments with skin. She is also working with external partners. Moreover, other substances such as senolytics are being developed and tested for their effect on ageing cells.4
The researchers from Ulm now want to drive forward the in vivo experiments with the substance CASIN, which inhibits the activity of the small Rho GTPase Cdc42 and rejuvenates old stem cells. According to Geiger, Cdc42, a key molecular switch in signal transduction processes, is not only important for the ageing of the immune system, but also for other tissues. The small protein, whose diverse physiological functions have not yet been fully explored, is a central switch that establishes order at many levels. Geiger explains that an unfunctional switch results in protein disorders, including epigenetic changes.
The upcoming experiments will attempt to break down the complexity and focus on individual tissues. This might also provide answers as to whether the immune system is a catalyst for ageing and whether ageing research could possibly just focus on a few tissues. Geiger and his group hope to come up with answers to these questions within five to ten years. They also hope that their findings will be able contribute to healthier ageing.