Product testing under real conditions
INSPIRE Living Lab: a bridge between industry and the clinic
Coming up with innovative ideas and products in the medical care area is highly relevant not only for the further development of Baden-Württemberg as a health region, but also for improving healthcare itself. However, bridging the gap between industry and the clinic often proves difficult. To drive this process forward, the INSPIRE Living Lab was opened at the University Medical Centre Mannheim in 2021. It is a place where start-ups and SMEs can test and improve their products in everyday clinical conditions.
At first glance, the INSPIRE Living Lab at the University Medical Centre Mannheim (UMM) looks like a normal hospital ward, yet nothing is the same as a conventional ward. Although the facility is part of the hospital's standard care services, it is fundamentally different from other UMM departments. The interdisciplinary urology and orthopaedics ward is the first ‘living laboratory’ in Germany. In addition to patients, clinical staff and doctors, visitors can also meet start-ups and SMEs here. The concept behind the ward: company founders and entrepreneurs who have developed a medical product or an idea for one have the opportunity to test, optimise and further develop their applications directly in everyday clinical conditions.
Facilitating everyday clinical routine through product testing under real conditions
 The interdisciplinary urology and orthopaedics ward opened in 2021 on the grounds of Mannheim University Hospital. © Mirjam Schleske / INSPIRE Living Lab
The interdisciplinary urology and orthopaedics ward opened in 2021 on the grounds of Mannheim University Hospital. © Mirjam Schleske / INSPIRE Living LabA product that is being tested is expected to be able to contribute to facilitating and improving daily routine in the clinic – this is the living lab’s key mission. And it has proved successful, as the system is beneficial for all involved: while the innovative technologies make the work of clinical staff easier, start-ups in particular benefit from the living laboratory settings. Much of the clinical data they need for obtaining approval for their products is generated directly under real conditions. This is fundamentally different from the usual test environments. In addition to collecting clinical data for approval, usability studies can also be conducted to analyse the usability of a product. Patients also benefit from the ward's modern technical equipment. The ward’s basic equipment includes a tablet computer at every bedside, which patients can use to communicate directly with nursing staff, among other things.
Following the introduction of the MDR and IVDR, the University Medical Centre Mannheim realised very quickly that more extensive clinical data collection would be required in future to prove the functionality and safety of medical devices. The INSPIRE Living Lab therefore provides a platform to collect data quickly and easily in a clinical context. The ward was opened in June 2021 after several months spent setting up a modern technical infrastructure. Funding for converting the hospital ward into the INSPIRE Living Lab was provided by the European Union, and the ward is now financially self-supporting. The project is part of the overarching INSPIRE network, which has the overall goal of driving forward technological transformation in healthcare. Significant support is also provided by the city of Mannheim’s business development department.
State-of-the-art technical infrastructure and expert knowledge
The INSPIRE Living Lab has developed a three-tier offer to drive forward technological transformation in hospital wards across Baden-Württemberg. Entrepreneurs looking to test a product in the special ward are invited to an initial meeting, free of charge, with Mirjam Schleske, the Living Lab’s organisational director. During the meeting, the two parties will prepare a checklist to define the goals and expectations for a Living Lab phase and clarify how the tests can be conducted meaningfully on the ward. Consultations with experts including physicians, clinical nursing staff or the hospital's IT department are also part of the offer. Many start-ups seek contact with physicians in particular to get feedback on the usability and relevance of their innovations.
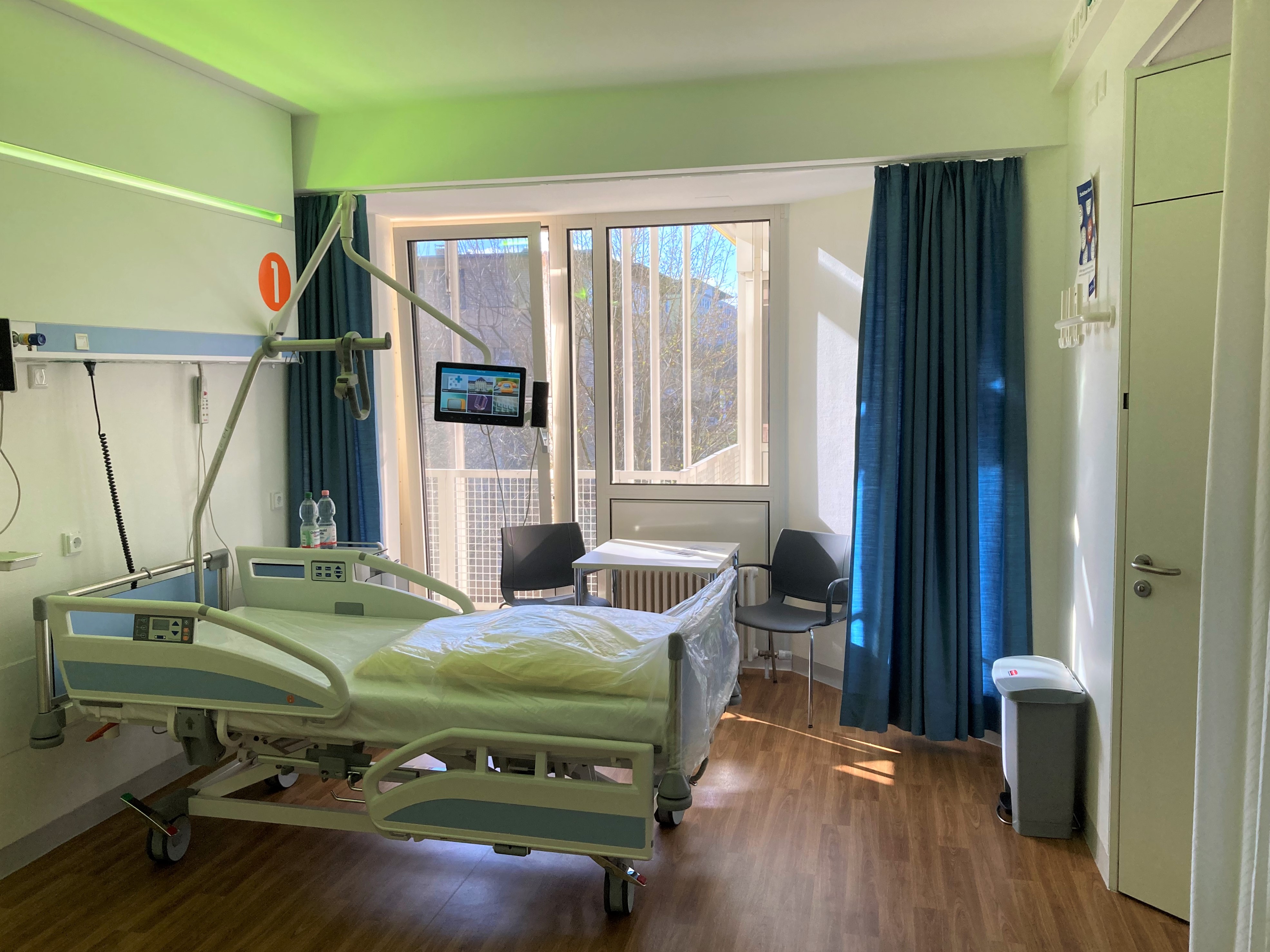 A patient room on the INSPIRE ward, equipped with tablet computers for the patient and the latest technical infrastructure required for bedside testing. © Mirjam Schleske / INSPIRE Living Lab
A patient room on the INSPIRE ward, equipped with tablet computers for the patient and the latest technical infrastructure required for bedside testing. © Mirjam Schleske / INSPIRE Living LabPractical implementation for the living lab phase can be designed in many different ways. The decisive factor here is the type of product that will be tested and its current development status. Whether the testing phase ends with technical documentation, the need for a clinical trial or a list of reference customers is one of the major issues that will be dealt with during the time at the INSPIRE Living Lab. Projects such as larger clinical trials or CE certifications are outsourced to partners. The organisational management of the INSPIRE Living Lab acts as a single point of contact for the experts and thus effectively supports start-ups during the entire testing phase.
The Living Lab's offer also includes access to the hospital's data platform. By exchanging data with the hospital information system, the data pool can be significantly increased, and this in turn facilitates product optimisation , for example when an app is being developed. In this context – as with all other tests on the ward – data protection-compliant security is the top priority. The Living Lab is particularly careful with tests that involve patients, because their well-being and successful recovery, as well as compliance with personal data protection rights, are paramount. Innovative applications involving patients require the provision of extensive information and explicit consent. With the patients’ consent, the data collected can also be used for clinical studies.
"The start-ups’ innovative ideas tend to be well received by patients, and many enjoy contributing to the further development of the innovations," says Schleske. Cliniserve, the first pilot project to be undertaken when the Living Lab opened, is considered a real Living Lab success. Cliniserve is an app aimed at relieving the burden on nursing staff through the adoption of digital solutions. Nurses are directly connected to patients on their ward via a smartphone or tablet computer and can prioritise nursing inquiries and thus optimise processes. The time saved is more time that nursing staff can then spend with patients.
Everyone benefits – especially patients
According to Schleske, it is no coincidence that the INSPIRE Living Lab is located on an orthopaedics and urology ward. Many people with oncological diseases are treated in urology, and urology wards are well suited for digital projects. Applications for patient management (e.g. monitoring symptoms) can be especially helpful in oncology. They can also help collect quality-of-life and symptom data from real-world care for quality assurance, including, for example, drug benefit assessments.
On the other hand, orthopaedics provides an excellent test scenario for sensor technology and applications related to rehabilitation measures. This makes both of these interdisciplinary areas particularly well suited as a living lab for digital health applications. In the best-case scenario, it might be possible to transfer these solutions to the entire hospital. The Living Lab is certainly pursuing a broader vision that envisages the entire patient journey taking place in a fully digitised hospital.
If there is one piece of advice Schleske would like to give start-ups and SMEs, it is to seek contact with the hospital as early as possible. Even during the idea development phase, initial dialogue with the hospital can be helpful and save time in further development: "Talking to the people who will later work with the product makes development much more efficient, as real needs in the clinic's daily routine can be addressed right from the start. And that's what the Living Lab is all about: implementing innovations quickly, effectively and safely in everyday hospital routine so that it can be optimised for everyone involved."