SolidCAR-T project
Modular ‘mini-factories’ for decentralised production of CAR T cells
Novel CAR T-cell therapies have proved to be promising therapeutic options for the treatment of acute leukaemias and lymphomas. Researchers from the Fraunhofer IPA in Stuttgart, the University Hospital Tübingen and the NMI in Reutlingen have joined forces in the SolidCAR-T project that aims to generate CAR T cells to combat solid tumours. This will also involve finding a way to produce these cells directly on site in the clinic using automated 'mini-factories', which would be significantly more cost-effective than current processes.
Different therapeutic approaches are used to treat cancer. The main ones are surgical interventions, chemotherapy or radiation therapy. Immunotherapeutic treatment strategies that use the patient’s immune system to fight cancer are also increasingly being used. These novel approaches are among the most promising.
CAR T-cell therapies are based on the way the body's own T lymphocytes function. The T lymphocytes are part of the body’s specific immune defence and their role is to eliminate infected or degenerated cells. In CAR T-cell therapy, the patient’s T lymphocytes are genetically modified in such a way that they form an artificial receptor on their surface, known as a CAR (chimeric antigen receptor), which helps them to recognise and eliminate cancer cells. This targeted form of therapy requires the molecular characterisation of an individual’s tumour, something that is done in the context of personalised medicine. CAR T-cell therapy belongs to the group of advanced therapy medicinal products (ATMPs) that open up new possibilities beyond just oncology.
CAR T-cell therapy: individual and complex
Since 2018, two CAR T-cell therapies have been approved in Germany, both directed against the CD19 antigen found on B cells, and used to treat B-cell leukaemias and lymphomas. They are currently produced manually in what is a complex procedure: T lymphocytes are removed from the patient by leukapheresis (separation and collection of the white blood cells), genetically modified using lentiviruses in the laboratory so that the CAR forms on their surface, and are then returned to the patient after multiplication.
One of the greatest challenges associated with ATMPs is the quality requirements for gene and cell therapeutics, which means that development and production is restricted to a few production centres only. This added to the often very long development times, major development risks and elaborate clinical trials makes treatment very expensive – a single dose currently costs around 275,000 euros. Price is therefore the reason why therapies are not yet widely available. Due to the high quality requirements and the dependence on a product-specific manufacturing licence, there are still no standardised manufacturing processes for ATMPs that would significantly reduce production costs.
‘Mini-factories’ for decentralised production
 Andreas Traube and Sarah Kleine-Wechelmann from Fraunhofer IPA coordinate the SolidCAR-T project and the development of the 'mini-factories'. © Fraunhofer IPA
Andreas Traube and Sarah Kleine-Wechelmann from Fraunhofer IPA coordinate the SolidCAR-T project and the development of the 'mini-factories'. © Fraunhofer IPAAn interdisciplinary consortium consisting of the Fraunhofer Institute for Manufacturing Engineering and Automation (IPA) in Stuttgart, the University Hospital Tübingen (UKT) and the Natural and Medical Sciences Institute (NMI) in Reutlingen is seeking to reproduce the entire development chain of an ATMP based on a selected indication: bile duct cancer. This chain would include production of the viral vectors, process development and automation of the manufacturing process, new in vitro test methods for demonstrating the efficacy of the produced therapeutics and inline quality control. "Personalised medicine requires new production methods. We believe it is much better to decentralise production to the site where the patients are, in other words, the clinic itself," explains Andreas Traube, head of Fraunhofer IPA’s Laboratory Automation and Bioproduction Technology department and responsible for the automation of a sub-process in the project.
The SolidCAR-T project, funded by the Baden-Württemberg Ministry of Economic Affairs, Labour and Tourism as part of the Forum Health Region BW, therefore aims to develop scalable modular production units, quasi ‘mini-factories’, that can produce high quality CAR T cells locally in a standardised, automated process. Production of the therapeutic agent thus becomes more technology-based, more widely available and adaptable to local needs.
The central component of the ‘mini-factories’ are standardised cassettes into which the T lymphocytes are transferred directly after leukapheresis. Once inside the cassettes, the T lymphocytes pass through the different stations of the production process. Each cassette contains biochambers with different prevailing conditions depending on the production step. Biochambers with different conditions are necessary because "transduction with lentiviruses requires cell environment conditions that are different from the expansion phase during which the cells multiply," explains Sarah Kleine-Wechelmann, who is coordinating the project at Fraunhofer IPA alongside Traube. Standardised, sterile connections ensure the highest levels of hygiene. "As the individual systems are closed, the safety requirements do not have to be so strict and we will be able to produce in a cleanroom with lower classification standards, which will significantly reduce costs and the work required." Furthermore, the modular design enables scalability and adaptability, paving the way for parallel processing of different product classes in one infrastructure.
CAR T cells to treat solid bile duct cancers
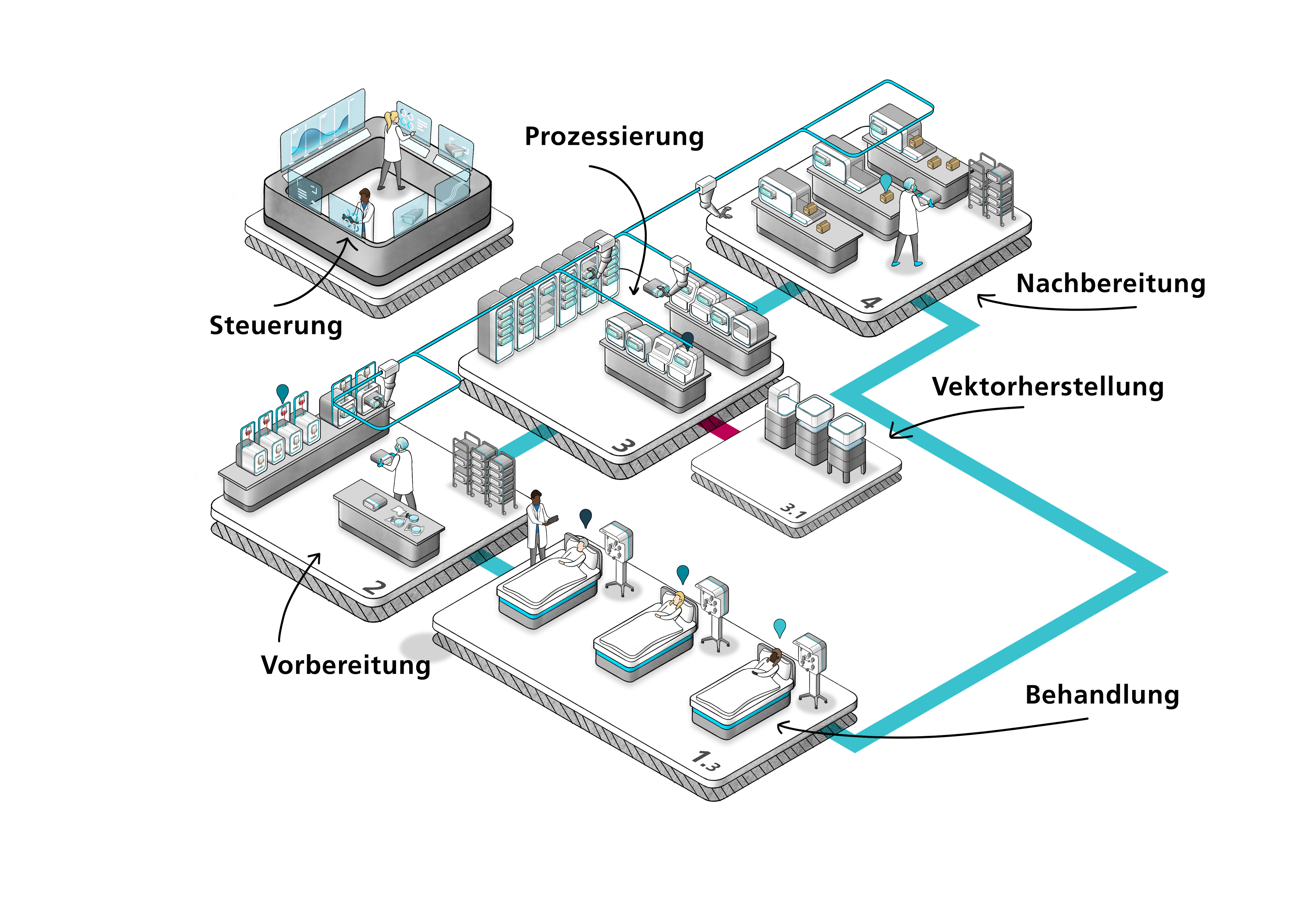 Illustration of the modular production concept, i.e. the ‘mini-factory’, which is aimed at enabling more cost-effective production of CAR T cells on a decentralised basis in clinics. © Fraunhofer IPA
Illustration of the modular production concept, i.e. the ‘mini-factory’, which is aimed at enabling more cost-effective production of CAR T cells on a decentralised basis in clinics. © Fraunhofer IPAThe UKT has a high level of expertise in the production and use of cellular therapies. As part of the SolidCAR-T project, Dr. Christian Seitz’ research group in the Department of Paediatrics and Adolescent Medicine is working with Prof. Dr. Nisar Malek’s Internal Medicine I department to establish and optimise the manufacturing processes of CAR T cells for treating bile duct cancer. In contrast to B-cell tumours, bile duct cancers are solid tissue tumours that are usually diagnosed relatively late, at an advanced stage that makes them difficult to treat.
The core task of the NMI under the direction of Prof. Dr. Katja Schenke-Layland is the quality assurance of cell products. In addition, patient-derived tumour models in combination with organ-on-a-chip systems are used in the laboratory model to simulate what happens in patients. This contributes to quality assurance and can also predict the effectiveness of the cell products even before patients receive the drug. "The SolidCAR-T project is an opportunity for further developing precision medicine," Schenke-Layland points out.
The parallel development of the manufacturing process and production machinery within a production system forms the core of the SolidCAR-T project, which is receiving a total of 4 million euros in funding.
Process monitoring improves quality assurance
Another special feature that will be an improvement on the current process is the continuous monitoring of the individual steps, thus enabling process parameters to be adapted to the needs of the cells and consequently improve quality. "Our major goal is to be able to ensure a smooth transfer of the production units to new locations and other cell systems while maintaining the same quality," says Traube, describing the future scenario. "In order to be able to do this, parameter monitoring is an important prerequisite," she adds. SolidCAR-T is also intended to lay the foundation for a centre for the production of individualised cell therapeutics, which will enable the technology to be transferred to industrial partners in the long term. It is estimated that around one in ten cancer patients are eligible for personalised drug therapy. So there is a major requirement for increased production capacities to be able manufacture affordable products.