Signal mechanisms in inflammatory processes
Newly discovered substances activate the inflammasome in macrophages
Low-grade chronic inflammation caused by components of the innate immune system may increase the risk of developing a variety of diseases in the long term. In their search for the signalling mechanisms underlying these inflammatory processes, Prof. Dr. Olaf Groß' research group at the Freiburg University Medical Centre discovered new active substances with immunostimulatory properties that may open up additional possibilities in cancer immunotherapy.
 Prof. Dr. Olaf Groß has discovered new active substances with immunostimulatory properties. © CIBSS, University of Freiburg
Prof. Dr. Olaf Groß has discovered new active substances with immunostimulatory properties. © CIBSS, University of Freiburg"We are not made for this world that we have built for ourselves. Evolution has not adapted us to this lifestyle," is how Prof. Dr. Olaf Groß describes the situation in which we live, where increasing age is often characterised by diseases of civilisation such as type 2 diabetes, arteriosclerosis or gout, as well as cancer. Many of these diseases caused by chronic, low-grade immune activity, typically in the form of inflammation, which displays no symptoms for decades and causes hidden damage to the tissue. It is only as we grow older that the consequences become visible. Drivers of the inflammatory reactions include macrophages, which are special scavenger cells of the innate immune system. Together with natural killer cells, granulocytes, mast cells and other immune cells, they directly ward off harmful substances and pathogens with the help of inherited recognition mechanisms. This is in contrast to the acquired immune defence mediated by T and B lymphocytes, which generates a tailor-made immune response, including the formation of antibodies. The cells of innate and acquired immune system communicate with each other and with other parts of the body via soluble messenger substances, so-called cytokines.
The NLRP3 inflammasome causes sterile inflammation
Macrophages secrete pro-inflammatory cytokines after they are activated. These attract other immune cells and activate them so that invaders or cancer cells can be destroyed in a targeted way. The cytokines of the interleukin-1 (IL-1) family are particularly effective, above all IL-1β. The release of IL-1β is controlled by the inflammasome, a multiprotein complex in the cytosol of macrophages, which is composed of several copies of a sensor molecule, the adapter protein ASC, and the inactive precursor of the enzyme caspase-1. The complex forms only after the cell has recognised danger signals emanating from pathogens or irritants. It subsequently activates caspase-1, which in turn generates mature IL-1β and cleaves gasdermin-D, a protein that subsequently forms pores in the cell membrane through which the cytokines are released.
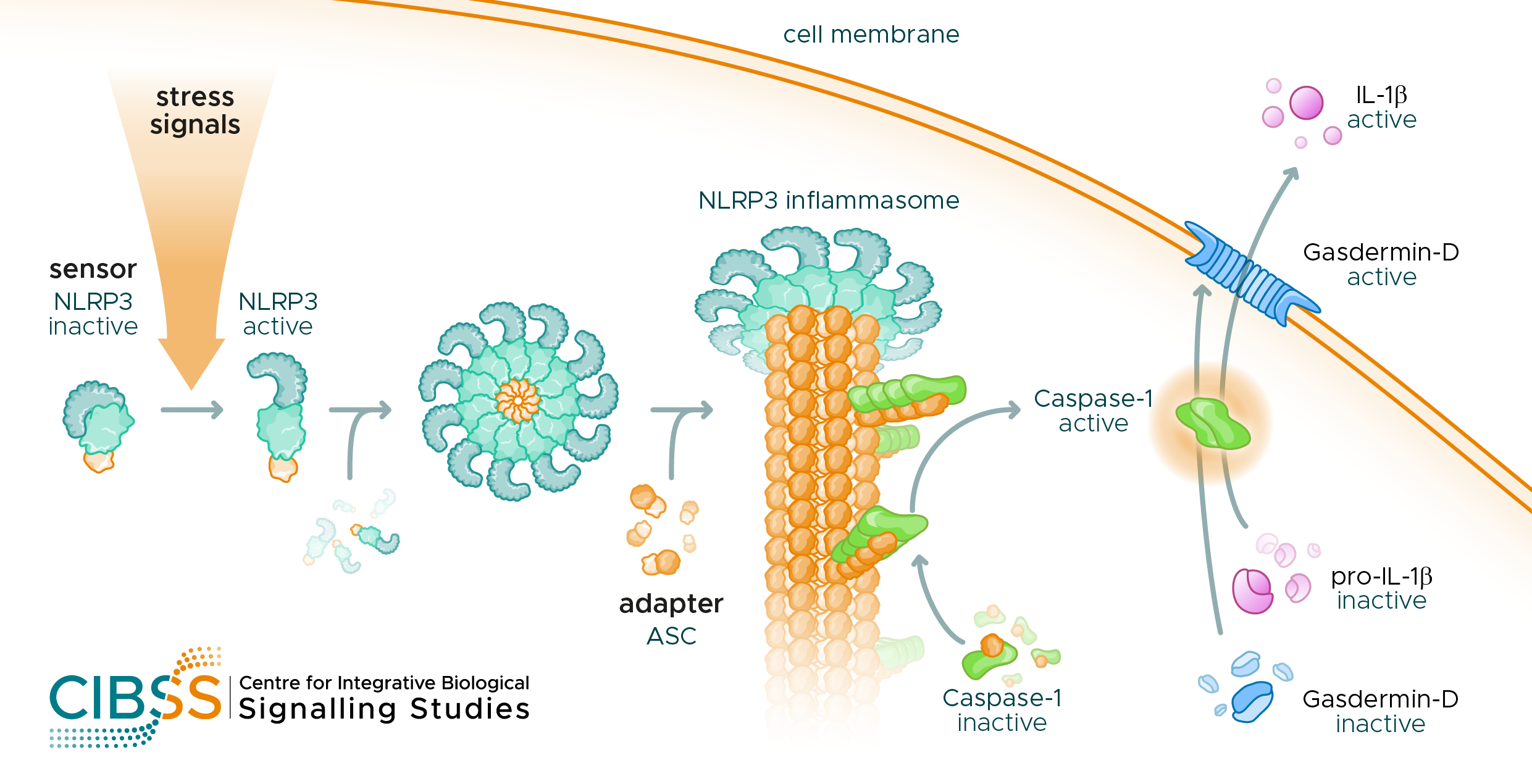 Mechanism of interleukin-1β (IL-1β) release in macrophages. After stress signals are detected by the NLRP3 sensor molecule, the NLRP3 inflammasome complex assembles and activates caspase-1, which generates mature IL-1β and cleaves gasdermin-D. The latter then forms pores in the cell membrane through which IL-1β is released. © CIBSS, University of Freiburg
Mechanism of interleukin-1β (IL-1β) release in macrophages. After stress signals are detected by the NLRP3 sensor molecule, the NLRP3 inflammasome complex assembles and activates caspase-1, which generates mature IL-1β and cleaves gasdermin-D. The latter then forms pores in the cell membrane through which IL-1β is released. © CIBSS, University of FreiburgDepending on the sensor molecule different inflammasome types exist. The NLRP3 inflammasome, which is responsible for chronic inflammation, reacts primarily to irritants and not pathogens, causing so-called sterile inflammation. Asbestos, for example, triggers inflammation of the lung tissue in this way, which can lead to lung cancer in the long term. Inflammation due to uric acid or cholesterol crystals, on the other hand, intensifies the symptoms and consequences of gout or arteriosclerosis. The NLRP3 inflammasome is hugely important medically because its continuous activation is linked with the development of many diseases. Normally, immune responses are finely balanced; they must be strong enough to generate a good defence, but they must not be stronger or last longer than necessary in order to avoid damaging the body. "We do not understand why the immune system is unable to switch off these low-grade inflammations," explains Groß, who has been working intensively on the NLRP3 inflammasome since his postdoctoral period and has led a research group at the Institute of Neuropathology at the Freiburg University Medical Centre since 2017.
New NLRP3 inflammasome activators found
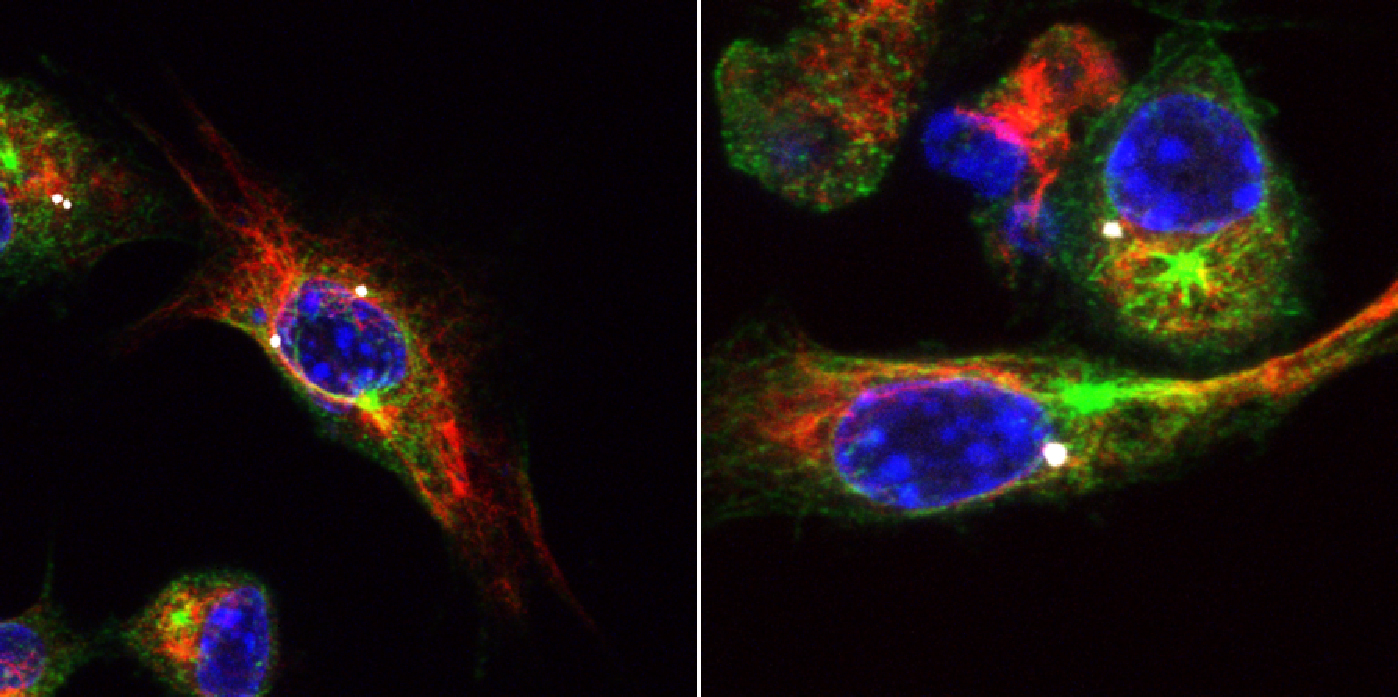 Macrophages after inflammasome activation. Fluorescence microscope image showing activated inflammasome (white), cell nuclei (blue) and the structural proteins tubulin (green) and β actin (red) © CIBSS, University of Freiburg
Macrophages after inflammasome activation. Fluorescence microscope image showing activated inflammasome (white), cell nuclei (blue) and the structural proteins tubulin (green) and β actin (red) © CIBSS, University of FreiburgSo far, there are no approved drugs that can inhibit the NLRP3 inflammasome, partly because the signalling chains that trigger its activation are still unknown. "My lab's strategy is to study the mechanism of activators in order to better understand what NLRP3 ‘sees’. Once we understand this mechanistically, we will be able to develop better inhibitors," explains the basic researcher. In the search for active substances that activate the NLRP3 inflammasome, Groß’ team has developed a new test procedure and used it to examine a collection of 50,000 chemical substances. Thanks to this compound screening, the scientists were able to identify several classes of molecules that showed significantly better potency and also specificity than previously known activators. One of the major tasks over the next few years is to decipher the signalling mechanism, including direct interaction partners, in order to create a basis for the development of inhibitors. This will mainly take place within the framework of the Centre for Integrative Biological Signalling Studies (CIBSS) Cluster of Excellence at the University of Freiburg, and in cooperation with other CIBSS groups.
Potential application in cancer therapy
The activators that were identified can possibly also be used to directly support cancer treatments. The microenvironment of malignant tumours contains a wide variety of immune cells whose function can, however, be manipulated by the tumour cells. These send signals that can switch off immune cells or, in the case of macrophages, even reprogramme them so that they acquire tumour-promoting properties. Tumour-associated macrophages are therefore an important target in cancer therapy. The specific activation of the NLRP3 inflammasome with the help of the newly discovered active substances could put the immune system back on track.
With future therapeutic use in mind, Groß has now filed a patent application. He explains why: "Non-patented research is basically worthless for the industry. If the patent rights are not secured, then there is very little motivation to further develop basic knowledge towards the market." Building on previous funding from the European Research Council (ERC), the researcher has now been awarded ERC Proof of Concept funding to explore the commercial potential of his team’s research results, among other things. Over the next few months, the researchers will investigate the tolerability of the NLRP3 inflammasome activators and whether they can improve existing cancer therapies or possibly also the immune response to vaccines. These preclinical data increase the market potential and thus create a basis for attracting investors. There are also plans to further develop the substances chemically in order to increase their efficacy and specificity and thus their tolerability. The targeted activation of macrophages of the innate immune system could open up new possibilities in many therapeutic areas.