Assay to support the diagnosis of autoimmune diseases
Tracking down pathogenic immune complexes
Soluble complexes of antibodies and their target structures circulating in the blood can trigger serious systemic inflammations. Dr. Philipp Kolb and Haizhang Chen from the Institute of Virology at the Freiburg University Medical Centre have developed a sensitive, cell-based test system for detecting these immune complexes. The system can be used to diagnose systemic autoimmune diseases, and also severe cases of COVID-19.
Antibodies are an essential part of our acquired immune response and are normally produced in response to contact with foreign bodies, such as those found primarily on and in pathogens. They bind specifically to their target, thereby attracting immune cells that eliminate the foreign substances. For reasons that are still largely unknown, antibodies directed against the body's own cells, tissues or organs occasionally develop. Autoimmune reactions of this kind lead to chronic inflammation and, in the long term, destroy healthy tissue. This either selectively affects individual organs, such as the pancreas in diabetes mellitus and nerve fibres in multiple sclerosis, or the autoantibodies cause systemic diseases such as systemic lupus erythematosus (SLE, antibodies against nuclear components) or rheumatoid arthritis (RA, chronic joint inflammation) that can affect the whole body.
Soluble immune complexes cause systemic inflammation
 The test system developed by Dr. Philipp Kolb and Haizhang Chen can detect minute amounts of soluble immune complexes circulating in the blood. © University of Freiburg
The test system developed by Dr. Philipp Kolb and Haizhang Chen can detect minute amounts of soluble immune complexes circulating in the blood. © University of FreiburgImmune complexes are formed after an antibody has bound to its target structure, i.e. the antigen. Depending on where the antigen is located, the immune complexes are either immobilised on a cell surface or occur in soluble form as sICs (soluble immune complexes; antigen-antibody complexes that circulate in the blood and lymph). The size and composition of the immune complexes depend on how many antibody-binding sites (epitopes) the antigen has, and on the ratio of the components to each other. In most cases, the immune complexes occur locally and are quickly eliminated by the cells of the immune system. However, both systemic autoimmune diseases and chronic viral or bacterial infections are often associated with soluble immune complexes circulating in the blood. These can deposit anywhere in the body and not only cause local reactions and tissue damage, but also life-threatening inflammation of the entire organism due to the immune system being constantly active. Until recently, sICs have been quite difficult to detect, so the clinical benefit has been considered low.
"With our assay, it is not only possible to detect the smallest quantities of soluble immune complexes, but also to predict their effect on the immune system",1) said Dr. Philipp Kolb, group leader at the Institute of Virology at the Freiburg University Medical Centre, referring to the advantages of his new test system. "Initial investigations on patient samples show a direct correlation between our values and the clinical score, i.e. disease severity." The method was developed largely by PhD student Haizhang Chen, and uses living cells to emulate the immune system's response.
Fcγ receptors for detecting sICs
Immunoglobulins (IGs), the technical term for antibody molecules, have a y-shaped structure and each possesses two identical antigen-binding sites. These are located at the ends of the two ‘arms’ and formed of highly variable regions. This is why each individual can produce a large number of antibodies with a wide variety of antigen recognition properties. In contrast, the ‘tail’ of the immunoglobulins is constant and only varies between the five antibody classes. This region, known as the Fc region, interacts with specific recognition molecules, the Fc receptors, located on the surface of immune cells. The majority of immunoglobulins found in the blood belong to class G (IgG) and are recognised by Fcγ receptors, which are found, for example, on the surface of phagocytes and natural killer cells. However, a simple interaction between Fcγ receptors and IgG molecules is not sufficient to activate immune cells. Neighbouring Fcγs need to be connected with each other; something that happens when they simultaneously bind different antibodies attached to the same antigen. This triggers a signalling cascade within the immune cell, leading either to the release of messenger and cytotoxic substances or to the uptake of the immune complexes through phagocytosis.
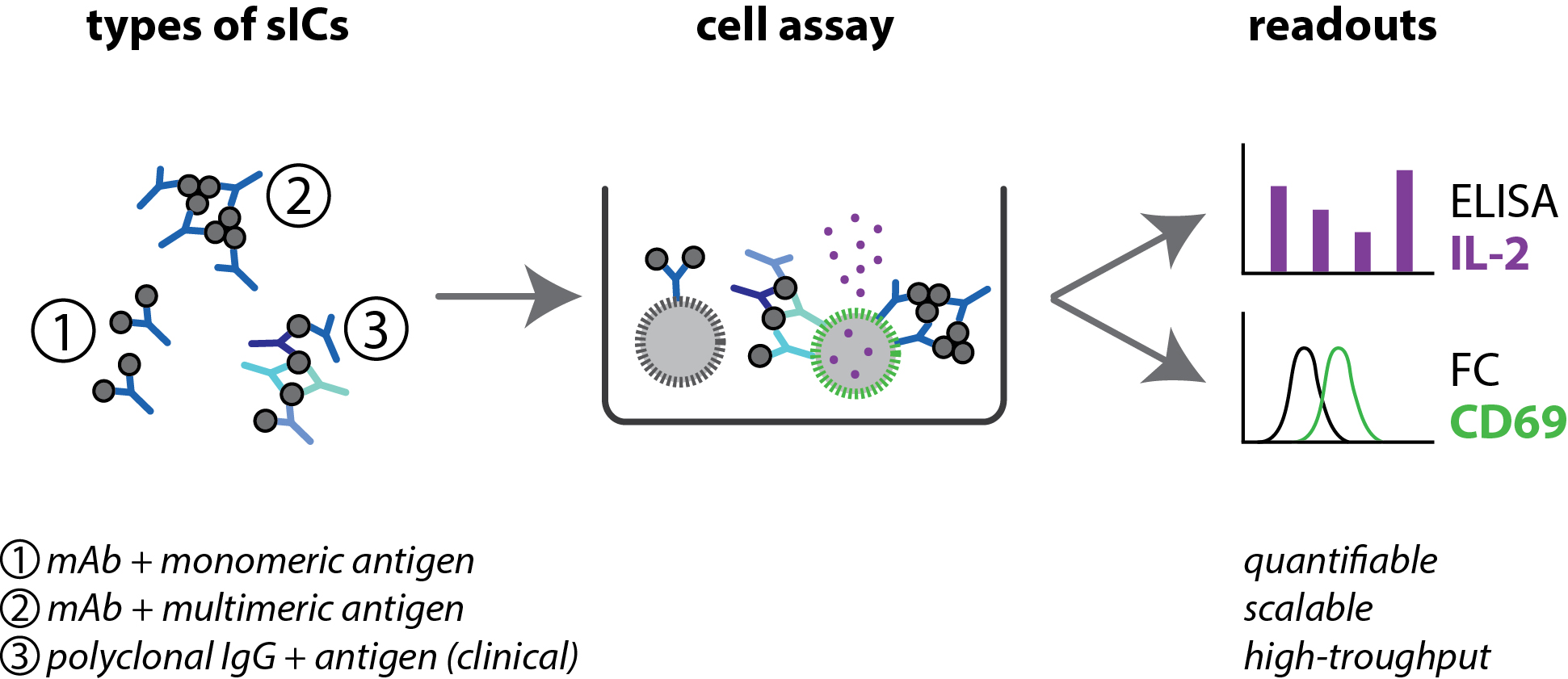 Soluble immune complexes (sICs) can vary in size depending on whether the antigen has only one antibody-binding site/epitope (1), multiple identical epitopes (2) or different epitopes recognised by different antibodies (3). Only the binding of sICs containing multiple antibodies activates reporter cells, releases interleukin-2 (IL-2, purple) and forms CD69 (green). Both signals can be quantitatively assessed using either the ELISA technique or flow cytometry (FC). Source: https://doi.org/10.15252/emmm.202114182, Philipp Kolb, CC-BY 4.0 (https://creativecommons.org/licenses/by/4.0/deed.de), modified by R. Menßen-Franz.
Soluble immune complexes (sICs) can vary in size depending on whether the antigen has only one antibody-binding site/epitope (1), multiple identical epitopes (2) or different epitopes recognised by different antibodies (3). Only the binding of sICs containing multiple antibodies activates reporter cells, releases interleukin-2 (IL-2, purple) and forms CD69 (green). Both signals can be quantitatively assessed using either the ELISA technique or flow cytometry (FC). Source: https://doi.org/10.15252/emmm.202114182, Philipp Kolb, CC-BY 4.0 (https://creativecommons.org/licenses/by/4.0/deed.de), modified by R. Menßen-Franz.The researchers use precisely these cellular signalling pathways to detect sICs in the blood. They established a model system for primary immune cells by producing a mouse reporter cell line that carries a human Fcγ receptor on its surface.
The recognition of sICs activates the cells and causes them to form the receptor molecule CD69 on their surface and release the soluble messenger substance interleukin-2.
Both effects can be evaluated independently and allow direct conclusions to be drawn about the immune-stimulating factors. Kolb explains: "Our system specifically emulates the activation of the Fcγ receptor. We measure the binding of immune complexes to the receptor, and we can also see precisely to what extent an immune cell reacts to it." This enabled the researchers to show that both the quantity and the size of sICs is crucial. A large complex elicits a significantly stronger cell response than several smaller ones. "Our detection method is about a thousand times more sensitive than common biochemical tests because of the signal amplification that occurs due to the cellular cascade that has been set in motion. We can detect the inflammatory factors at a very early stage and thus predict the possibility of autoimmune attacks. In addition, the system is stable and scalable. This means it can measure single samples as well as hundreds of samples at a time." Mass sampling will likely prove to be of great practical benefit to testing clinics.
Test indicates severity of the disease
To substantiate the clinical relevance of the assay, Chen analysed the serum of 25 SLE sufferers who form autoantibodies against DNA or DNA-bound proteins and who have sICs circulating in their blood. He was able to show a direct correlation between the activity of the samples in the test system and the severity of the disease. When using the test system to investigate joint fluid samples from people with rheumatoid arthritis, Heidelberg researchers confirmed that sICs also play a role in inflammation in this case.2)
Doctors have known for some time that severe COVID-19 cases are very similar to autoimmune diseases such as SLE and that lupus drugs work well to treat symptoms of COVID-19. Studies using the test system now show that the serum of affected individuals contains high levels of sICs.3) Again, the severity of the disease correlates with the reactivity of the sICs. Interestingly, however, the sICs do not contain coronavirus antigens, but seem to be composed of autoantibodies against a wide variety of the body's own tissues. A pilot study is currently underway with severely ill patients to determine whether the course of therapy can be tracked via the presence of the sICs in the patients’ blood and thus allow a conclusion to be drawn about the degree of cure. "If one can detect when the inflammatory factors have disappeared from the blood, the administration of medication that is often hard to tolerate can be minimised," explains Kolb.
Interdisciplinary success
"The assay is the result of interdisciplinary cooperation," reports Prof. Dr. Hartmut Hengel, head of the Institute of Virology at the Freiburg University Medical Centre. "We are virologists and are actually investigating how the cytomegalovirus can escape the Fc receptors’ immune control. We were asked by rheumatologists to develop a cell-based system to measure the activation of other receptors, but we were unsuccessful. So Mr. Kolb simply investigated the rheumatology samples using an Fc receptor assay which he had adapted for this purpose and observed a strong response." Thanks to interdisciplinary cooperation, a candidate with a great deal of potential has thus emerged from an unfunded side project. The researchers plan to get the test system certified in the near future to enable its regular application in clinical diagnostics.