Peptides – diverse molecules of life
Peptides exist in all organisms, wherever there are cells. The range of their physiological functions is huge. Biologically active peptides can act as hormones, neurotransmitters, growth factors as well as toxins and antibiotics. This is what makes them highly interesting drug leads. They are used for the treatment of autoimmune diseases, cancer and other diseases. Despite some drawbacks, peptides are gaining in importance as candidates for drugs and fuelling interest in research into natural and synthetic peptides.
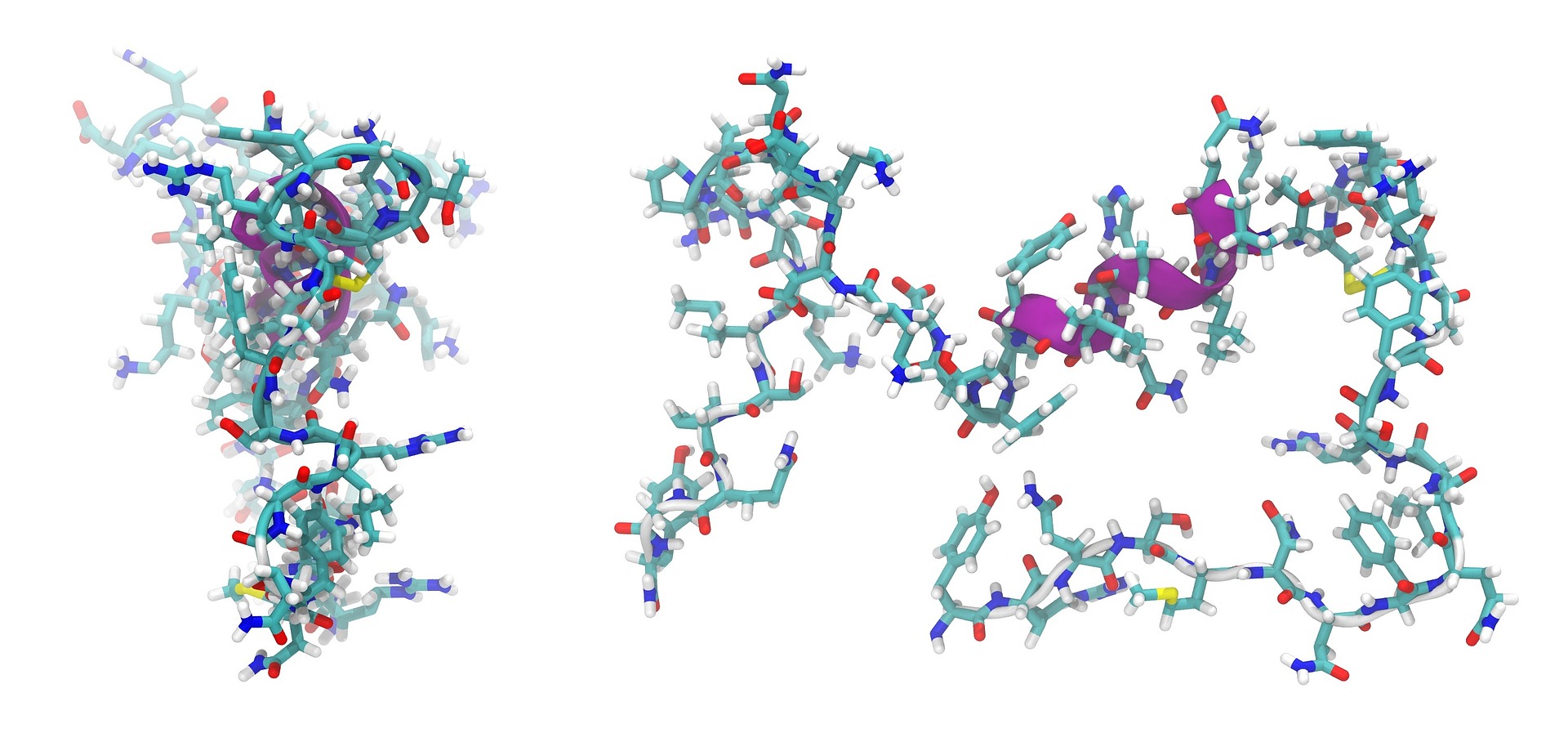 Adrenomedullin is a peptide consisting of 52 amino acids. On the left side, the peptide is shown in its native form, while the secondary structure is shown on the right side. © WikimediaImages / pixabay
Adrenomedullin is a peptide consisting of 52 amino acids. On the left side, the peptide is shown in its native form, while the secondary structure is shown on the right side. © WikimediaImages / pixabayPeptides consist of amino acids, but are smaller than proteins. Some researchers classify peptides over 50 amino acids long as proteins, whereas others set a benchmark at 100 amino acids. The individual building blocks are joined by relatively rigid peptide bonds, forming a chain of amino acids. These bonds are formed when the carboxyl group (COO-) of one amino acid reacts with the amino (NH3+) group of another, thus releasing a water molecule. In addition to peptide bonds, disulphide bonds can exist within and between peptide chains, as can ester and thioester bonds. Tanja Weil from the University of Ulm reports that most peptides are linear chains of amino acids, but that there are also cyclic and branched peptides.
Peptides can be produced using different pathways
Natural peptides can consist of proteinogenic and non-proteinogenic amino acids. In order to keep track of their tremendous variation, peptides are grouped according to whether they are produced by the ribosome machinery or not. Eukaryotic organisms use ribosomes for synthesizing peptides. The protein synthesis machinery translates mRNA into protein by polymerising the 20 proteinogenic amino acids based on the mRNA sequence. The resulting proteins are subsequently cleaved into smaller peptides by proteolytic enzymes. Like proteins, ribosomal peptides also undergo posttranslational modification (e. g. phosphorylation, glycosylation, sulphation, hydroxylation and the formation of disulphide bridges).
Bacteria and fungi are able to produce short peptides in different steps involving different enzymes. In addition to proteinogenic amino acids, bacterial and fungal peptides can also consist of non-proteinogenic L- and D-type amino acids. It has been shown that the stability of peptides increases with the number of D-amino acids. This makes them particularly interesting for the pharmaceutical industry. Many lead structures are derived from such bacterial peptides.
Fungal and bacterial peptides are synthesized by a modular multi-enzyme complex (nonribosomal peptide synthetase, NRPS). Each NRPS can synthesize only one type of peptide. Around 1,200 nonribosomal peptides (as of 7/2013) have been discovered in 247 organisms (https://www.gesundheitsindustrie-bw.debioinfo.lifl.fr/norine).
Huge obstacles: extraction and isolation
Before peptides can be synthesized using chemical and/or recombinant tools, they need to be extracted and isolated, a process that often fails as peptide mixtures can degrade when they are removed from their biological environment. Researchers who want to copy natural peptides need outstanding analytical skills as well as access to techniques such as mass spectrometry, liquid chromatography (HPLC), tandem spectrometry and magnetic resonance in order to glean information on the peptide’s structure, mass, etc. Many analysis techniques are required to achieve a complete picture. There is more to the detective work, as Tanja Weil calls it, especially when the synthesized peptide does not have the biological activity it is expected to have. If these initial hurdles are successfully overcome, the scientists are able to synthesize a peptide of interest.
Focus is now on longer peptides
The term peptide goes back to the German chemist and Nobel Prize winner (1902) Emil Fischer who succeeded in synthesizing the first dipeptide in 1901. With the establishment of solid-phase synthesis, (R. M. Merrifield was awarded the Nobel Prize in Chemistry in 1984 for this achievement) peptide research received a boost as it then became possible to chemically synthesize peptides on a solid matrix. Merrifield’s discovery opened up the possibility of producing larger quantities and purer substances. The solid-phase synthesis process was later automated and peptide synthesizers were placed on the market. In addition, peptides can also be produced using recombinant technologies.
Modern peptide chemistry is focused on ways to synthesize particularly long peptides as peptide synthesizers are technically only able to synthesize peptides up to 30 amino acids long. Tanja Weil explains that the construction of longer peptides involves the ligation of several short peptides into longer polypeptides.
Peptides have always been attractive for chemists for their role as multifunctional building blocks. The peptides’ reactive groups (carboxyl and amino groups) can be used for joining amino acid monomers (polymerization). The side chains of amino acids (with the exception of glycine which has only a hydrogen substituent as its side chain) can be equipped with functional (protecting, activating) groups. The ability to attach protective groups to side chains enables the production of macromolecules of a defined sequence and length.
The company PEPperPRINT from Heidelberg uses a unique way for producing peptide microarrays; the peptides are synthesized on-chip using amino acid microparticles and a custom colour laser printer.
Peptides as drugs
The number of peptide drugs is increasing dramatically. It is estimated that hundreds of peptide-based drug candidates are currently in the clinical phase and an even larger number in preclinical drug development. As all organisms are able to produce antimicrobial peptides, researchers around the world are looking for new effective antimicrobial drugs in a broad range of animals, plants and fungi, including in animals like toads, spiders and snakes. In the meantime, peptides with antiobiotic effect, so-called microzines (see Zschüttig et al.), have been identified in E. coli and other bacteria.
The newly founded Ulm Centre for Peptide Pharmaceuticals (UPEP) is particularly focused on human peptides. UPEP researchers are screening the human peptidome for the presence of bioactive and immunomodulatory substances. They have already discovered human peptides that minimize the infectiousness of HIV.
A peptide-based vaccine against renal cancer developed by the Tübingen-based biotech company immatics biotechnologies GmbH is one of the most prominent examples of therapeutic peptides. The company is currently testing the vaccine in a phase III clinical trial. Peptides are also used in the field of medical technology. A number of research groups are focusing on the development of implants coated with antimicrobial peptides or with peptides that increase the implants’ biocompatibility.
In addition to the field of therapeutics, peptides are also of major interest in the production of synthetic high-performance materials. Researchers at the Institute of Technical Biochemistry in Stuttgart are focused on composites consisting of peptides and inorganic components. Such constructs are of major interest for the construction of electronic components and devices.
Walter Pytlik – 31.03.2014
© BIOPRO Baden-Württemberg GmbH
Literature:
Königshoff M, Brandenburger T. Kurzlehrbuch Biochemie. Stuttgart/New York: Thieme 2012 (3., rev. version)
Caboche S, Leclère V, Pupin M, Kucherov G, Jacques P: Diversity of monomers in nonribosomal peptides: towards the prediction of origin and biological activity. J Bacteriol 2010;192(19), 5143-50, doi: 10.1128/JB.00315-10
Venoms for Health, EU project of the 7th FRP (2011-2015): https://www.gesundheitsindustrie-bw.dewww.venomics.eu
Zschüttig A, Zimmermann K., Blom J, Goesmann A, Pöhlmann Ch, Gunzer F: Identification and Characterization of Microcin S, a New Antibacterial Peptide Produced by Probiotic Escherichia coli G3/10, March 30, 2012, DOI: 10.1371/journal.pone.003335 (PLoS ONE), https://www.gesundheitsindustrie-bw.dewww.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0033351
Emil Fischer - Biographical: https://www.gesundheitsindustrie-bw.dewww.nobelprize.org/nobel_prizes/chemistry/laureates/1902/fischer-bio.html
The Antimicrobial Peptide Database: https://www.gesundheitsindustrie-bw.deaps.unmc.edu/AP/main.php
Problems associated with the design of peptide drugs
Although peptides have been used for the treatment of human diseases for many years, the administration of peptides still poses a huge problem. Peptides are degraded rapidly in the gastrointestinal tract and therefore need to be administered subcutaneously or intravenously. In addition, they are relatively quickly cleared from the blood circulation by the liver and kidneys. A considerable amount of research therefore focuses on overcoming the challenges presented by oral administration, including the development of new drug delivery forms and the modification of peptide structures that increase the molecules’ stability, for example. However, as it is relatively easy to synthesize peptides and as peptides can target many physiological processes, they are of huge interest for the pharmaceutical industry despite the aforementioned challenges.
KS