Bakteriocins
New antibiotic alternatives from soil bacteria
Bacteria are not the greatest of friends among themselves: many release antimicrobial substances into their environment in order to gain an advantage in their ecological niche. Researchers at the University of Ulm are making use of such bacteriocins by creating a genetically modified soil bacterium that can be used as a biotechnological platform organism to produce alternatives to antibiotics in pure form and in large quantities. The fact this uses waste materials also saves on resources.
Humans and animals like to flex their muscles to drive unwelcome competitors out of their territory, but microorganisms entirely lack this possibility. However, what they do have is something much more alarming: bacteriocins. These small, antimicrobial peptides are produced by a variety of bacteria and can kill or at least inhibit the growth of other bacteria. Their biological role is to ensure the survival of the bacteria that have produced them, thus allowing them to survive and propagate in their ecological niche.
But bacteriocins are also important for us as humans, and because they act against bacteria, they are already widely used as preservatives in food and animal feed. Nisin, for example, whose E number is E 234. The lactic acid bacterium Lactococcus lactis uses this antimicrobial substance to create pores in biomembranes of competing bacteria, which kills them. However, bacteriocins are completely harmless to humans and animals. They are natural constituents of milk, for example, and are quickly degraded in human and animal digestive tracts.
Using bacteriocins to avoid antibiotic resistance
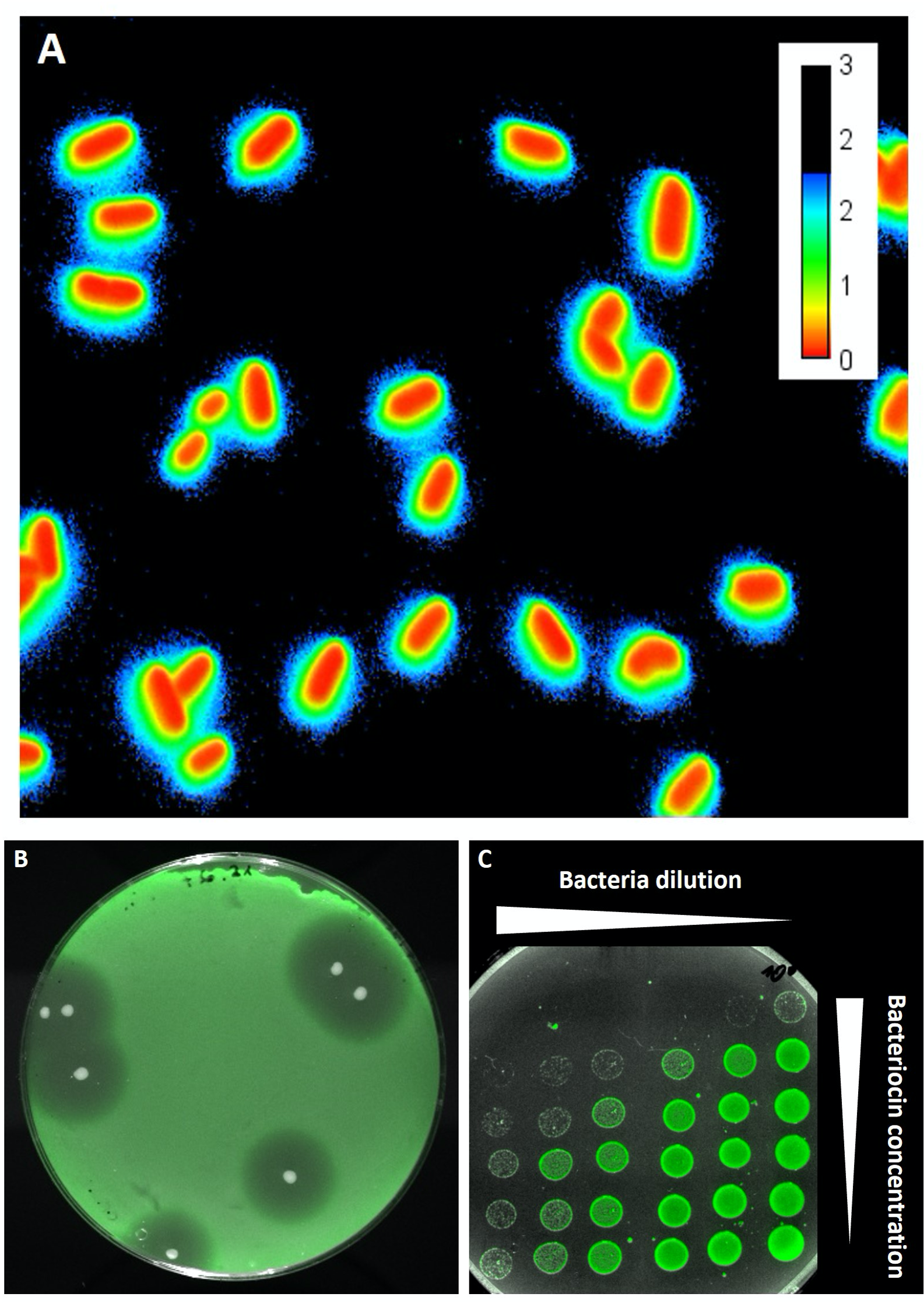 The fluorescence microscope images show individual bacteria (A) and cultures on agar plates (B, C). A) Bacteriocins can be detected using fluorescent sensor bacteria, which glow blue-green in the outer area as soon as they come into contact with the antimicrobial sub-stance. B) Around the white colonies of the bacteriocin producers, inhibition zones can be seen in the lawn of the sensor bacteria. C). The growth of the sensor bacteria is inhibited – how strongly depends on the dilution and bacteriocin concentration. © Christian Riedel / University of Ulm
The fluorescence microscope images show individual bacteria (A) and cultures on agar plates (B, C). A) Bacteriocins can be detected using fluorescent sensor bacteria, which glow blue-green in the outer area as soon as they come into contact with the antimicrobial sub-stance. B) Around the white colonies of the bacteriocin producers, inhibition zones can be seen in the lawn of the sensor bacteria. C). The growth of the sensor bacteria is inhibited – how strongly depends on the dilution and bacteriocin concentration. © Christian Riedel / University of UlmBacteriocins have also been viewed for quite some time as having medical potential: they are regarded as promising alternatives to common antibiotics. Due to the fact that antibiotics have been used excessively and abusively in recent years, especially in animal husbandry, many bacteria have become resistant to what was once a very effective medical weapon. According to the World Health Organisation (WHO), bacterial resistance is currently one of the greatest threats to our health.1)
In fact, in recent years, a number of bacteriocins have been obtained from a wide variety of bacterial sources and show promising effects against clinically relevant pathogens. However, their practical application has so far been hampered by several factors: they have to be produced in complex fermentation processes using natural bacteria, which require complex and expensive culture media. At best, this results in semi-purified preparations or raw ferments that still have to go through a complex purification process for medical use. In addition, the procedure has high energy requirements, making it far too complicated and unprofitable for any applications other than food preservation.
At least until recently. Now a solution is within reach. Researchers at the Institute of Microbiology and Biotechnology at the University of Ulm have shown that it is biotechnologically possible to produce a highly effective antimicrobial peptide using the industrial platform organism Corynebacterium glutamicum – a non-pathogenic soil bacterium – in such a way that peptide production can also be transferred to an industrial scale.2 ) "Working with European partners in the iFermenter project, we have developed a production process that can be used to produce recombinant bacteriocins in an efficient and resource-saving way on an industrial scale," says Prof. Dr. Christian Riedel, who is leading the research work in Ulm. "The first peptide we produced is pediocin PA-1, and it is particularly effective against listeria bacteria, such as those that occur in raw milk and can trigger dangerous disease symptoms. Now we want to extend the process gradually to other peptide pharmaceuticals and expand the product portfolio – not only to produce antibiotic alternatives, but also hormones or antiviral substances, for example."
Bacteriocin production as a modular system
 Researchers Dr. Oliver Goldbeck, Prof. Dr. Christian Riedel, Dominique N. Desef and Prof. Dr. Bernhard J. Eikmanns (from the left) have created a recombinant soil bacterium that can efficiently produce bacteriocins (Dominik Weixler, who is also involved in the research, does not appear in the photo). © Lisanne Wolters / University of Ulm
Researchers Dr. Oliver Goldbeck, Prof. Dr. Christian Riedel, Dominique N. Desef and Prof. Dr. Bernhard J. Eikmanns (from the left) have created a recombinant soil bacterium that can efficiently produce bacteriocins (Dominik Weixler, who is also involved in the research, does not appear in the photo). © Lisanne Wolters / University of UlmThe researchers genetically modified corynebacteria to produce pediocin PA-1: for this purpose, the gene responsible for bacteriocin biosynthesis was introduced into the bacteria along with the genes required for protein modification and transport. Additional artificial genes give the bacteria the ability to use waste streams as substrate. "Waste such as lignocellulosic hydrolysates from the wood industry, for example, contain many interesting sugars," explains Riedel. "These sugars can then be used by ‘our’ bacteria to produce higher-value products such as bacteriocins."
The overall goal is to create a modular system: the genetic environment for the basic components will remain the same; only the gene modules required for the conversion of certain wood waste products and the production of certain substances will have to be exchanged. This would allow the modified corynebacteria to produce different substances and use different kinds of waste as substrate.
More products are under development
As regards pediocin PA-1, the international iFermenter team is currently working on transferring the process from laboratory scale to large scale for future industrial production: "Together with our Belgian partners, we are currently testing this in a pre-industrial 150-litre scale," says Professor Riedel. "In parallel, we are working on other, more complex products, such as nisin."
One challenge is the fact that the soil bacteria, thanks to their artificial genes, are supposed to produce substances that have an antimicrobial effect. However, these must not harm the bacterial producer itself. In the case of pediocin, this is not a problem: Corynebacterium glutamicum has no bacteriocin receptors. However, it’s a different story for nisin, which is why the experts have a trick up their sleeve: an inactive precursor is first formed, which is subsequently activated in a second step.
Tests in creams and bandages coming up
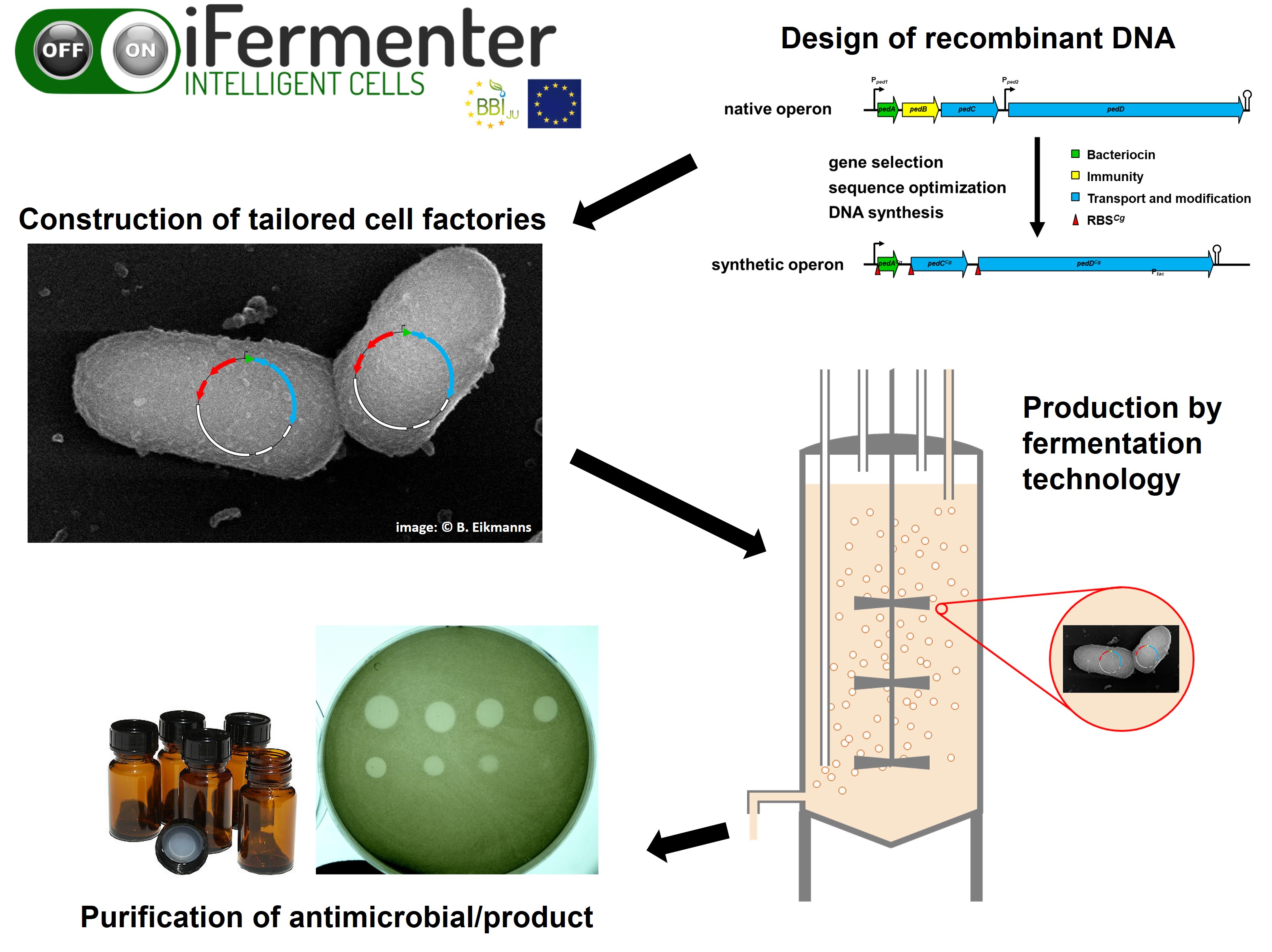 The iFermenter research project established the recombinant production of bacteriocins with the help of the model organism Corynebacterium glutamicum in order to make the bacteriocins available for medical use. © Christian Riedel / University of Ulm
The iFermenter research project established the recombinant production of bacteriocins with the help of the model organism Corynebacterium glutamicum in order to make the bacteriocins available for medical use. © Christian Riedel / University of UlmThe bacteriocins will have to pass the first practical test in antimicrobial creams and dressing materials. "A joint application with the medical device manufacturer Hartmann is underway," says Riedel. The substances are not suitable for oral use as they would be rendered ineffective in the gastrointestinal tract. Moreover, even administered intravenously, the bacteriocins would be eliminated relatively quickly by the immune system. Research activities are therefore concentrated on external application, for example on the skin and mucous membranes. However, packaging them into capsules would enable them to be used as probiotics that alter the microbiota. In any case, the researchers from Ulm have a lot of work to do in the near future: in addition to further developing what they have already achieved, they are looking to expand the search for antibiotic alternatives in all possible directions – screening strain collections, analysing soil samples and, as part of basic research, searching for new substances. And then, of course, commercialise the developed process as quickly as possible.
iFermenter project
The research work on bacteriocins in Ulm is part of the international iFermenter research project, which receives EU funding of around 5.25 million euros within the framework of Horizon 2020. iFermenter is coordinated by the Norwegian University of Science and Technology in Trondheim. The joint project is developing intelligent bioprocess technologies to produce antimicrobial peptides from wood industry waste streams. The research project aims not only to add greater value to waste streams that contain sugar, but also to make production processes cheaper and more resource-efficient.