COVIC-19 clinical trial
Convalescent plasma for COVID-19 therapy: clinical trial should bring clarity
Does it help to treat COVID-19 sufferers with antibodies from people who have recovered from the disease? It seems an obvious idea and has been tested thousands of times. However, there is not yet enough evidence to prove the clinical efficacy of treating COVID-19 patients with convalescent plasma. Existing study data are inconclusive and hard to compare. The transfusion physician Prof. Dr. Hubert Schrezenmeier from Ulm is planning to carry out a follow-up clinical trial to find the missing evidence.
This follow-up randomised controlled clinical trial (RCT), funded by the German government, will be commencing very soon. Its aim is to untangle the mountain of complex method-related data and provide clarity on the clinical effectiveness of this therapeutic option in the fight against the pandemic.
The coronavirus pandemic has set in motion an unprecedented development of therapeutics against COVID-19 (634 projects worldwide)1). In summer 2021, after several wake-up calls, the German government put together a 150-million-euro package for the development of application-oriented therapeutics.2) Funding will only be provided for drugs that are produced in sufficient quantities and used clinically – something that is guaranteed at the GRC Blood Donation Service Baden-Württemberg – Hesse, one of the largest alliances of GRC blood donation services in Germany, with large manufacturing capacities for blood products and plasma.
Info box: What is convalescent plasma?
Convalescent plasma (CP) is blood plasma from persons who have successfully recovered from an infectious disease, in this case caused by the coronavirus SARS-CoV-2, and who have developed immunity to the pathogen in question. CP contains antibodies that bind to and neutralise the pathogen, thereby helping the immune system to fight infection. CP is considered a safe therapeutic agent, even in combination with other COVID-19 therapies, as demonstrated by the information available in large haemovigilance databases. Emil von Behring discovered the principle of plasma therapy in the late 19th century and it has since been used for successfully treating numerous infectious diseases. Convalescent plasma is administered either by transfusion or injection. Blood plasma is obtained using a well-established method called plasmapheresis.
First trial provides indications for follow-up trial
 Prof. Dr. Hubert Schrezenmeier © GRC BW-Hesse / IKT University of Ulm
Prof. Dr. Hubert Schrezenmeier © GRC BW-Hesse / IKT University of UlmResearchers led by Prof. Dr. Hubert Schrezenmeier conducted Germany's first clinical trial3) involving CP as COVID-19 therapy. In this trial, CP therapy was compared with the best available supportive treatment in 105 patients with severe COVID-19. The trial did not achieve its objectives (prolonged survival and clinical improvement in disease severity at 21 days in the overall cohort). It did, however, provide evidence for a dose effect. The researchers found that high-dose CP may in fact have an effect when high concentrations of virus-neutralising antibodies are present in the plasma.
The upcoming follow-up clinical trial called COVIC-19 (Efficacy of early transfusion of convalescent plasma with very high antibody concentrations in severely ill patients with COVID-19 as a model for early therapy options in pandemic situations caused by new pathogens) will attempt to demonstrate clinical efficacy with a very early, high-dose therapy in a selected, particularly vulnerable patient group. The control group will be treated with the best available standard therapy.
Administering early and high-dose convalescent plasma improves outcome
In the new clinical trial, only very high-dose plasmas with antibody titres that are higher than the SARS-CoV-2 antibody concentrations administered to the high-dose group in the first clinical trial will be applied. The plasma antibody concentration will be at least ten times higher than that used in the previous clinical trial. The trial, called CAPSID, found that CP should be given very early on to hospitalised patients who are at a clinically stable stage, i.e. within three to five days after the onset of symptoms. 4) The ALAMA prognosis score5), which reliably determines the risk of severe COVID-19 disease, will serve as a guide. The second patient group to be recruited will involve people who are immunocompromised due to congenital or acquired disease or who are undergoing immunosuppressive therapy. Schrezenmeier says that these people are known to have a poor prognosis and respond poorly to vaccination.
The aim of the new clinical trial is to prevent severe COVID-19 in these high-risk patients. "We would consider it a success if the condition of a patient who has been treated with CP does not worsen," explains Schrezenmeier, who heads up the Institute for Transfusion Medicine and Immunogenetics (IKT) in Ulm.
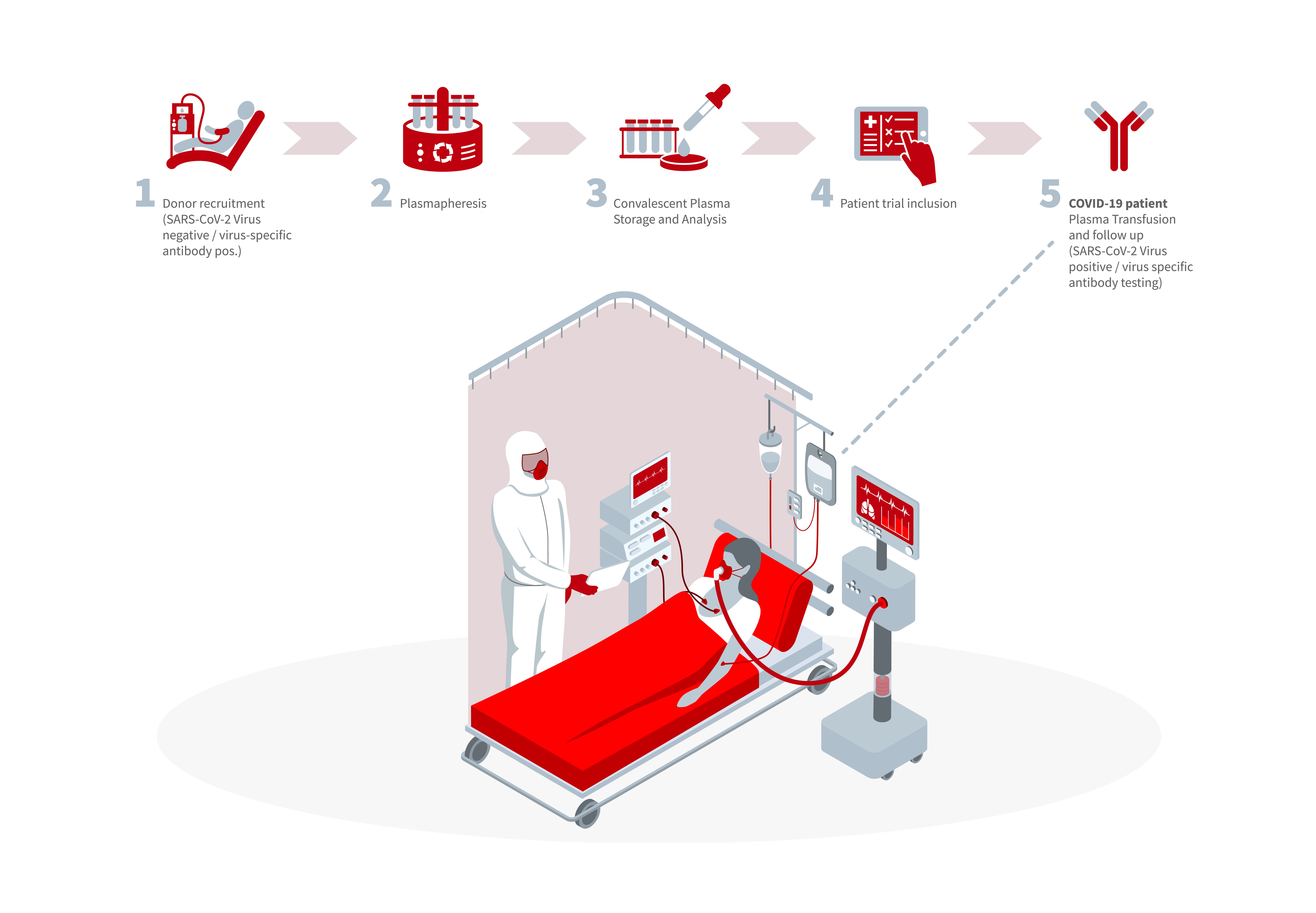 singleimage.big_view Schematic showing how the clinical trial will be performed.
Clinical trial flow chart: (1) recruitment of donors (SARS-CoV-2 virus negative / virus-specific antibodies positive), (2) plasmapheresis: collection of blood plasma, (3) convalescent plasma: analysis of plasma; (4) decision on inclusion in the patient trial, (5) COVID-19 patients: plasma transfusion and follow-up (SARS-CoV-2 virus positive / virus-specific antibody testing).
© GRC BW-Hesse / IKT University of Ulm
singleimage.big_view Schematic showing how the clinical trial will be performed.
Clinical trial flow chart: (1) recruitment of donors (SARS-CoV-2 virus negative / virus-specific antibodies positive), (2) plasmapheresis: collection of blood plasma, (3) convalescent plasma: analysis of plasma; (4) decision on inclusion in the patient trial, (5) COVID-19 patients: plasma transfusion and follow-up (SARS-CoV-2 virus positive / virus-specific antibody testing).
© GRC BW-Hesse / IKT University of Ulm
Suitable donor plasma is important
The selection of suitable donor plasma, which should have a high antibody titre and have developed an immunologically broad range of antibodies, is of decisive importance. In principle, patients to be included in the clinical trial will be given CP from donors with the same blood group. In addition, the distribution of blood groups in the donor group will also roughly correspond to that of the patient group.
As we enter the third year of the coronavirus pandemic, the COVIC-19 clinical trial is making use of an enormous increase in knowledge, which is key to a better understanding of CP therapy. For example, important basic principles on the development of antibodies after vaccination have been incorporated into studies: certain patient groups respond poorly to vaccination and require therapeutic help, despite high vaccination rates. Studies have shown – and according to Schrezenmeier's observations, a consensus is emerging in the scientific community – that people who are likely to have the highest antibody titres are those who have been immunised twice, i.e. who have been infected with the SARS-CoV-2 virus and also vaccinated – regardless of which of the two occurred first. It is precisely these ‘superimmunised’ donors with very high antibody concentrations that the researchers want to recruit.
Virus variants are part of the new clinical trial
 Blood plasma - its typical yellowish colour comes from the fact that it lacks red blood cells. © GRC BW-Hesse / IKT University of Ulm
Blood plasma - its typical yellowish colour comes from the fact that it lacks red blood cells. © GRC BW-Hesse / IKT University of UlmVariants of the virus – currently the rapidly spreading variant known as Omicron – are a challenge for researchers. The first CP clinical trial generation from spring/summer 2020 did not contain antibodies against virus variants. There was simply no information available on the virus variants that the patients were infected with. Therefore, nor was it possible to make a statement on whether the effectiveness of CP depends on the virus variant in the donor or in the recipient. The new generation of clinical trials now includes the investigation of virus variants. The IKT has developed a number of such tests, including whole-genome sequencing of SARS-CoV-2, and is conducting many investigations. Amongst other things, it is carrying out studies on variant-specific neutralisation in cooperation with the Institute of Molecular Virology at the Ulm University Medical Centre.
The design of the clinical trial, submitted in June 2021, not only provides for accompanying studies that will focus on characterising the antibodies and the virus, but also collecting precise details on the donors who are recruited. Thanks to new contacts through the coordinating EU project SUPPORT-E (SUPPORTing high quality evaluation of Covid-19 convalescent plasma throughout Europe)6), the clinical trial will also be conducted in two other countries (UK and France).
Unlike mono- and biclonal antibodies, whose antiviral effect can be completely lost if the viral variants against which they are directed develop resistance to them, polyclonal antibodies such as those present in CP are immune to a complete loss of effect due to cross-neutralisation. "At most, the effect only weakens gradually because the immune response achieved by CP is based on a spectrum of antibodies with slightly different epitopes," says Schrezenmeier. The working group was able to show that the CP of vaccinated people who have recovered from the disease also had an effect in COVID-19 patients infected with the new Omicron variant. 7)
The great advantage of CP is its flexibility
However, immune evasion can also occur with CP if a new virus variant is very different from that contained in the CP, for example when the CP comes from a donor who recovered from a completely different variant. Schrezenmeier is convinced that CP has a big advantage over monoclonal antibodies: "We can react much faster and motivate people who were infected with the new virus variant to donate CP. This makes it possible to adapt the CP system as quickly as possible so we always use CPs containing antibodies of virus variants that are circulating in the population at a given time."
The variants will be investigated in detail to find out whether CP therapies can be improved, for example by taking into consideration virus variants. This rapidly adaptive CP system can become a model for other pandemic situations involving viral pathogens where antibody defence plays a major role in combating disease. COVIC-19 thus has two goals: on the one hand, to contribute to the optimisation of COVID-19 therapy, and on the other, to develop a model for CP therapies for future pandemics that takes advantage of all the lessons learned so far.