Hummingbird Diagnostics GmbH
The great potential of blood-based microRNA analyses
"The early bird catches the worm", is an apt description of what motivates Hummingbird Diagnostics GmbH from Heidelberg. The medium-sized biotechnology company analyses special biomarkers in blood, so-called microRNAs, in order to diagnose diseases at an early stage and to be able to make forecasts about the course of the disease and the success of therapy.
Alongside proteins, carbohydrates and fats, nucleic acids are the fourth major group of biomolecules. In the form of deoxyribonucleic acids (DNA), they serve to store genetic information and are present in humans as long, double-stranded filaments tightly packed in the cell nucleus. Depending on the cell type, different sections of the DNA are transcribed. During this process, the corresponding DNA sequence is copied into single-stranded ribonucleic acid (RNA), which then passes from the nucleus into the cytoplasm. Only about two percent of RNA molecules contain the blueprint for proteins (mRNA); the remaining transcripts are either components of the protein synthesis machinery (rRNA, tRNA) or have regulatory functions. The latter include short microRNAs (miRNAs), which are usually composed of only 21 to 23 nucleotide building blocks. By attaching to regulatory mRNA regions, they influence the translation of mRNA into proteins and thus control a variety of cellular processes.
miRNAs as novel biomarkers
In many diseases, the biological processes within a cell are out of balance, which is reflected in an altered expression of miRNAs. The miRNAs, which are easily accessible and very stable in peripheral blood, thus open up new, non-invasive diagnostic possibilities. "microRNAs are wonderful biomarkers," enthuses Jochen Kohlhaas, CEO of Hummingbird Diagnostics. "As master regulators of gene expression, microRNAs play an essential role in altered health states - not just in cancer, cardiac or neurodegenerative diseases, but also in ageing. Although there are many research approaches, the analysis and assessment of miRNAs have proven difficult in practice with the technologies that are currently available. This is where robust, standardised methods are essential."
Development of a robust platform
 Jochen Kohlhaas is the founder and CEO of Hummingbird Diagnostics, a Heidelberg-based biotechnology company that specialises in the analysis of microRNAs for early disease detection and therapy prediction. © Hummingbird Diagnostics GmbH
Jochen Kohlhaas is the founder and CEO of Hummingbird Diagnostics, a Heidelberg-based biotechnology company that specialises in the analysis of microRNAs for early disease detection and therapy prediction. © Hummingbird Diagnostics GmbHThe researchers at Hummingbird Diagnostics have first-hand experience of this. Back in 2011 - at that time still going by the name febit, or Comprehensive Biomarker Center (CBC) - the company analysed the miRNA profiles present in the whole blood of healthy and diseased individuals.1) The studies included subjects with various tumours, multiple sclerosis and heart disease. It revealed a variety of general disease-associated changes in the miRNA signature. By applying bioinformatics, it was also possible to identify individual disease-specific miRNA molecules. Based on these and other results, Hummingbird Diagnostics launched a study for the early detection of lung cancer in the USA, but this did not deliver the hoped-for results.
This was mainly due to the massive occurrence of so-called batch effects, i.e. influences of non-biological factors such as handling, laboratory conditions or material differences, on the result. "In 2019, we therefore went back to square one and adapted all work steps from sample collection and storage to processing and analysis, thus creating a stable platform," explains Kohlhaas. "In doing so, Hummingbird expended significant resources to implement a largely automated, pipetting-free workflow that is robust and reproducible. Using proprietary molecular biology methods, we have now succeeded in capturing more miRNAs than has ever been possible with whole blood analysis."
The improved technology is already delivering promising results in the ongoing study for the early diagnosis of lung cancer. In addition, Hummingbird Diagnostics is currently working on a test for the early detection of Alzheimer's disease, for which the company is receiving funding from the Alzheimer's Drug Discovery Foundation (ADDF).
Therapy prediction using miRisk
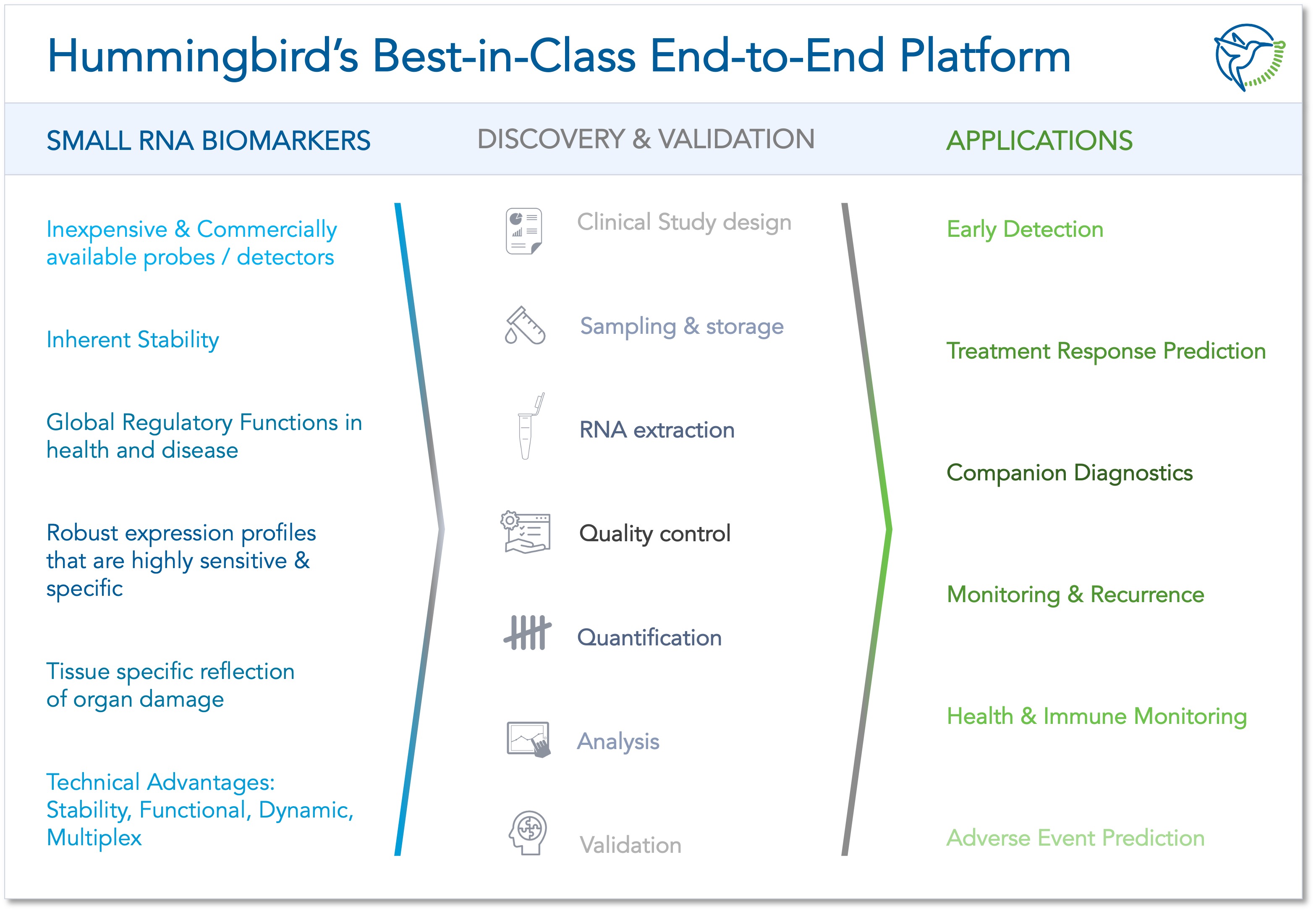 Hummingbird Diagnostics' miRNA platform enables a wide variety of miRNAs to be identified and evaluated for use in areas such as early disease detection, therapy prediction, companion diagnostics, or health status monitoring. © Hummingbird Diagnostics GmbH
Hummingbird Diagnostics' miRNA platform enables a wide variety of miRNAs to be identified and evaluated for use in areas such as early disease detection, therapy prediction, companion diagnostics, or health status monitoring. © Hummingbird Diagnostics GmbHHowever, the new platform has much more potential, as shown in the April 2022 publication in the Nature partner journal npj Precision Oncology.2) In the underlying study, Hummingbird Diagnostics uses miRNA analysis from whole blood to predict the likelihood of success of immunotherapies for advanced non-small cell lung cancer (NSCLC). At this stage, the tumour has already metastasised and conventional treatment methods offer little chance of a cure. Therefore, for some years now, novel active substances, so-called checkpoint inhibitors that activate the body's own immune system, have been used treating NSCLC.
Many tumour cells have a molecule known as PD-L1 (programmed death ligand 1) on their surface, with which they bind to and inhibit the PD-1 receptor of the immune cells. This blockade can be prevented by administering monoclonal antibodies, so that the tumour cells can be attacked and destroyed again. However, this is a relatively unspecific stimulation of the immune system, which is why these therapies can have severe side effects. In addition, they are only successful in about 30 percent of patients, even if tissue examinations have previously ensured that at least 50 percent of the tumour cells carry PD-L1 on their surface (tumour proportion score, TPS > 50).
Since immune cells circulate throughout the body in the blood, miRNA analyses from whole blood also allow conclusions to be drawn about a person's immune status. A team led by Dr. Bruno Steinkraus, CSO at Hummingbird Diagnostics, has now succeeded in using just five specific miRNAs to generate a risk score that can be used to predict the efficacy of therapy with checkpoint inhibitors in individuals with NSCLC. The predictive power of miRisk (miRNA risk score) is clearly superior to TPS in this regard. "If I can spare the 70 percent of those treated [for whom therapy doesn't work] the pain, and target those that it does help, that's a huge step forward," Kohlhaas elaborates. "Aside from that, the cost-effectiveness increases significantly."
Patient-friendly diagnostics
Another advantage of miRisk is that no surgical removal of tumour tissue is required and the method can therefore be used more widely. Moreover, it can also be used to identify patients who would have been excluded by the previous method, but who would probably benefit from the therapy.
It was only possible to develop the risk score through good cooperation with certain clinics in Germany. "With the Thoraxklinik Heidelberg and the LungenClinic Grosshansdorf, we had renowned cooperation partners with huge expertise in immunotherapies," Kohlhaas reports. Now, the predictive power of the method will be investigated in other cancer types that are treatable with checkpoint inhibitors and show whether miRisk is universally applicable. "We hope to launch miRisk in late 2023." Deployment will likely begin initially in the U.S. as a laboratory developed test (LDT) for use in the company's own diagnostic laboratory. In the long term, the development of a test kit is conceivable to make the technology easily accessible worldwide.
"Development is our real strength. We have a fantastic assay development team, great lab staff, and outstanding machine learning, artificial intelligence experts and bioinformaticians," says Kohlhaas, describing the company, which currently has just under 20 employees. "With our miRNA discovery platform, we have developed a tool that combines precise molecular biology measurements with deep machine learning and biological confirmation." These are ideal conditions for Hummingbird Diagnostics to develop further tests in the coming years to select disease- or therapy-relevant miRNAs at an early stage in other fields as well.