Intestinal peptide heals lung
Inhalation of intestinal hormone VIP helps against immunotherapy-induced pneumonia
If cancer patients develop pneumonitis, an inflammation of the lungs resulting from immunotherapy, their symptoms and restrictions in lung function can often only be alleviated with cortisone. Researchers led by Prof. Dr. Joachim Müller-Quernheim and Dr. Björn Frye from the Freiburg University Medical Centre have been able to cure a patient's pneumonitis by inhalation of a long-known neuropeptide.
Patients’ own defense systems are increasingly being used as allies in the treatment of various types of cancer. Immuno-oncology is considered a significant beacon of hope in medicine, due in part to the successes it has already achieved, for example, in treating melanoma. However, science faces huge challenges due to the numerous side effects of these therapies. If, for example, the lungs are attacked by the patient's own immune system as a result of immunotherapy, it is not uncommon to discontinue immunotherapy and slow down immune system reactions with cortisone. This is associated with the risk of the tumour growing again. Doctors at the Freiburg University Medical Centre have come up with a novel therapeutic approach involving the intestinal hormone VIP (vasoactive intestinal peptide), which was given by way of inhalation to a patient who had developed pneumonia as a consequence of immune therapy.
Prof. Dr. Joachim Müller-Quernheim and his team came up with the idea for the therapy involving VIP because of the histological and cytological similarities of immunotherapy-induced pneumonitis with the clinical picture of sarcoidosis. Sarcoidosis is an inflammatory disease affecting mostly the lungs and lymph glands, in which the immune system overreacts. A few years ago, Müller-Quernheim and his team were able to show that the inhalation of VIP reduced inflammation in the lungs, which in turn led to a significant improvement in the condition of those affected.
Checkpoints as natural immune system brakes
 Successful treatment of immunotherapy-induced pneumonia with an intestinal peptide: an idea of Dr. Björn Christian Frye (photo) and Prof. Dr. Joachim Müller-Quernheim © Freiburg University Medical Centre
Successful treatment of immunotherapy-induced pneumonia with an intestinal peptide: an idea of Dr. Björn Christian Frye (photo) and Prof. Dr. Joachim Müller-Quernheim © Freiburg University Medical CentreThe human immune system is faced with the enormous task of shielding the organism against invading pathogens and damaged (cancer) cells, while at the same time ensuring the safety of its own body tissues. The manifold mechanisms by which the immune system protects itself against autoimmunity without compromising an effective immune response against germs are enormously complex. By regulating the balance between pro-inflammatory and anti-inflammatory factors, the body attempts to establish a balance between autoreactive and immunosuppressive mechanisms. Inflammation is the immune system’s response to pathogens and other harmful stimuli, but it can also lead to serious damage. To promote self-tolerance, regulatory T cells attenuate inflammation. As degenerated body cells, cancer cells carry tumour-associated antigens on their surface that can be recognised by the immune system. However, many tumour types have strategies to disguise their malignant identity in order to remain undetected by the immune system. Some have features on the cell surface that attenuate the activity of effector cells.
These cancer cells activate so-called checkpoints on T cells, which act as control points to prevent the immune system from attacking its own cells, thus ensuring their survival. Immunotherapy with antibodies blocks the misuse of checkpoint receptors by the tumour, so that the tumour cells can be better recognised and destroyed.
When the immune system is unleashed
 Prof. Dr. Joachim Müller-Quernheim © Prof. Dr. Joachim Müller-Quernheim - private
Prof. Dr. Joachim Müller-Quernheim © Prof. Dr. Joachim Müller-Quernheim - privateImmunotherapy involving checkpoint inhibitors is a successful oncological treatment, but can also cause severe side effects. It not only removes the blockade of the immune system against cancer cells, but can also lead to the body's own cells being attacked, since protective mechanisms are inactivated. "Thus, lung damage associated with immunotherapy is observed in up to 20 percent of patients treated," the researcher says. "Lung structures and tissue are attacked, which impairs lung function and breathing."
Immune-related adverse events (irAEs,) are the consequence of an excessive autoimmune reaction as a result of immunotherapy. While many adverse events are considered acceptable due to the severity of the cancer, severe irAEs require the interruption or even discontinuation of immunotherapy. IrAE-related pneumonitis is treated with high-dose cortisone. On the one hand, the steroid hormone is an immunosuppressive and symptom-relieving drug, but on the other hand, due to its broad spectrum of activity, it can also cause many side effects. However, if cortisone is discontinued, pneumonia can also return.
An intestinal hormone cures the lungs
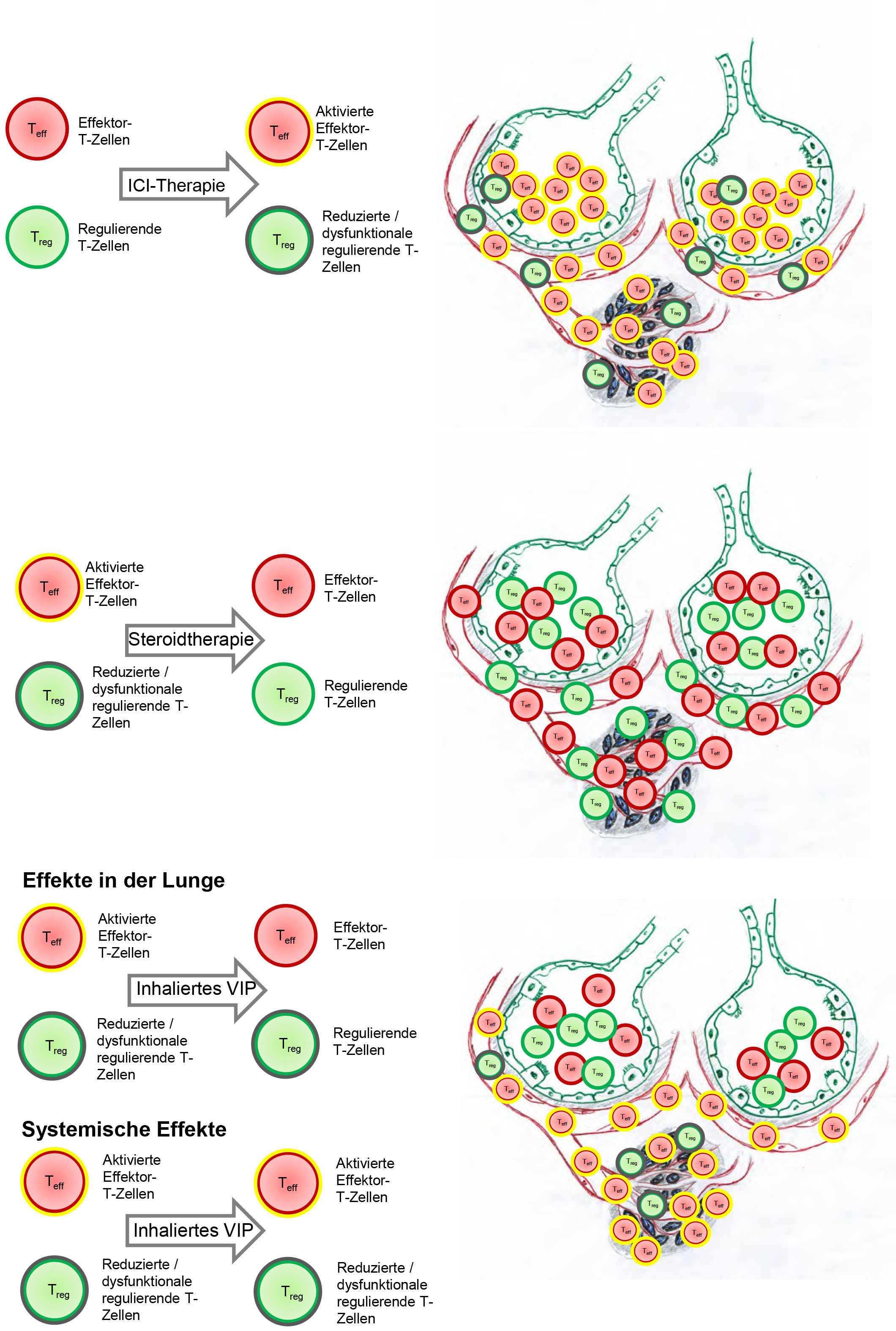 ICI (immune checkpoint inhibitor) immunotherapy activates effector T cells and conversely reduces the number of regulatory T cells in order to defeat the tumour (black cells). Pneumonia triggered by immunotherapy is treated with steroid therapy (cortisone) or with inhaled VIP (vasoactive intestinal peptide). VIP treatment attenuates the inflammation. © Dr. Björn Christian Frye - private
ICI (immune checkpoint inhibitor) immunotherapy activates effector T cells and conversely reduces the number of regulatory T cells in order to defeat the tumour (black cells). Pneumonia triggered by immunotherapy is treated with steroid therapy (cortisone) or with inhaled VIP (vasoactive intestinal peptide). VIP treatment attenuates the inflammation. © Dr. Björn Christian Frye - privateDue to the similarity of the inflammation of the lung in pneumonitis and sarcoidosis, the Freiburg scientists decided to apply the therapeutic approach that had been successful in sarcoidosis to a patient with pneumonitis in whom cortisone therapy did not lead to the desired outcome. The cause of sarcoidosis is still unclear, but there are indications that the patient's own immune system overreacts. The lungs are almost always affected, the disease causes clusters of inflamed tissue (granulomas) that impair lung function and lead to irritation of the throat and breathing difficulties. Cells from the lungs of sarcoidosis patients that have been examined have shown a high production of proinflammatory cytokines and few to zero regulatory T cells, as has also been observed in irAE pneumonitis. A study using an intestinal neuropeptide, the vasoactive intestinal peptide (VIP), will now investigate whether cough relief and an improvement in quality of life can be achieved in patients with sarcoidosis.
Many VIP receptors are found in the intestinal epithelium and the lungs as well as on leukocytes. Inhalation of aviptadil, a VIP formulation, attenuates the immune response. "When aviptadil is inhaled, large amounts of the inhibitor reach the lungs," says Müller-Quernheim. "Aviptadil does not reach the intestine at all, where it could cause adverse effects." After four weeks of treatment, doctors found that the release of TNF (tumour necrosis factor), the most important pro-inflammatory cytokine, had decreased significantly in the lungs. At the same time, an elevated number of regulatory T cells was observed. Both are essential to attenuate irAE pneumonitis in the lungs.
Anti-inflammatory neuropeptides
Müller-Quernheim's team also achieved success with aviptadil in a melanoma patient who had undergone immunotherapy with checkpoint inhibitors and subsequently developed pneumonitis. In this patient, the lung cells were also producing too much TNF and the level of regulatory T cells was too low. Aviptadil inhalation led to the reversal of both effects and the patient felt significantly better. It is expected that the effect of immunotherapy against the tumour will be maintained due to local application.
Over the past ten years, various neuropeptides that induce anti-inflammatory activities and play a major role in the self-tolerance of the immune system have been identified. Aviptadil and other neuropeptides interrupt the signalling pathways involved in the expression of multiple inflammatory mediators. This makes them interesting candidates for treating diseases where the immune system is out of balance. Use in COVID-19 treatment can also be envisaged. "The virus is usually eliminated quite well," says Müller-Quernheim. "But lung damage is a collateral effect of an excessive immune response, and this can be attenuated."