Organ-on-chip for the analysis of drug effects
New 3D cell platform allows continuous metabolic monitoring
Organ-on-chips systems are systems containing miniature tissues grown inside mircrofluidic chips. By integrating microsensors, researchers at the IMTEK Freiburg, together with the RWTH Aachen University Hospital, created a novel variant that allows the measurement of metabolic activity directly on site and in real time. This enables the rapid and detailed analysis of drug effects outside an organism.
In vitro cell culture systems are indispensable for investigating biological processes, but also for testing the effect and tolerability of drugs. Cells that do not grow in assemblies in the organism, such as blood and immune cells, are grown floating in nutrient solutions as a so-called suspension culture. However, most cell types grow adherently, i.e. they only multiply when they adhere to a surface. In conventional two-dimensional (2D) systems, these cells are cultured in nutrient medium on a glass or plastic support and form a single-cell layer known as a monolayer. However, this does not adequately reflect the situation in an organ, where cells grow in three-dimensional assemblies and interact with each other in all directions. For this reason, the findings obtained with the help of a 2D culture can only be transferred to a living organism in a limited way.
In recent years, more and more work has therefore been done to establish three-dimensional model systems in which the cells grow in the form of organ-like mini structures, so-called organoids. Since this promotes the formation of organ-typical properties, 3D cultures provide more clinically relevant results. For some time now, it has also been possible to grow the mini organs on special platforms that can imitate the microenvironment of a tissue with the help of small chambers and channels. In these organ-on-chip systems, the supply of nutrients and drugs can be precisely controlled and the influence on the growth of the cells can be studied in depth. However, the measurement of metabolic parameters has so far mainly been carried out using fluorescence staining and the removal of nutrient medium or cell material.
Integrated microsensors allow real-time measurement
 Dr. Andreas Weltin and Johannes Dornhof from the IMTEK in Freiburg developed a novel organ-on-chip system that can measure the metabolic activity of cells directly on the chip and in real time. © Andreas Weltin and Johannes Dornhof
Dr. Andreas Weltin and Johannes Dornhof from the IMTEK in Freiburg developed a novel organ-on-chip system that can measure the metabolic activity of cells directly on the chip and in real time. © Andreas Weltin and Johannes DornhofIn a project funded by the Baden-Württemberg Landesstiftung foundation, researchers at the University of Freiburg’s Department of Microsystems Engineering (IMTEK) have now succeeded in developing a novel organ-on-chip model that allows the metabolic activity of cells to be monitored locally and in real time. The special feature of the system is the integration of various microsensors, via which continuous monitoring is possible every second. "Determining oxygen levels, for example, must take place directly on the chip; pumping out the culture medium beforehand would falsify the data," explains Dr. Andreas Weltin, group leader in the Laboratory for Sensors. Since the consumption of the oxygen present in the medium correlates directly with aerobic cell respiration, i.e. the production of energy through the breakdown of glucose to carbon dioxide and water, it serves as a measure of cell vitality. "The measurement is only possible because, unlike many other organ-on-chip platforms, we use gas-tight materials," adds PhD student Johannes Dornhof, first author of the recently published work.1) The bottom of the 10×16 mm chip, for example, is made of glass, the walls of the channels and cell chambers are made of an epoxy-based photopolymer and the cover is made of Plexiglas.
This gas-tight system is especially important for cultivating and examining tumour cells. Due to their rapid growth, many solid tumours have oxygen-deficient (hypoxic) areas that promote metastasis and reduce response to therapies. Meaningful models must therefore emulate these conditions. "We can vary the oxygen content between air-like and highly hypoxic conditions," Weltin points out. In close cooperation with Dr. Jochen Maurer, the scientific head of Molecular Gynaecology at the Department of Gynaecology and Obstetrics at the RWTH Aachen University Hospital, the microsystems engineers transferred the cultivation of breast cancer stem cells established by the biologist to the chip. The cells are introduced into the growth chambers in a gel and supplied with nutrients via microchannels running along the sides. Thanks to microfluidics, the movement of the small amounts of fluid in the channels can be actively controlled, whereas the transport of substances within the cell chambers takes place by diffusion.
Live analysis of the energy metabolism
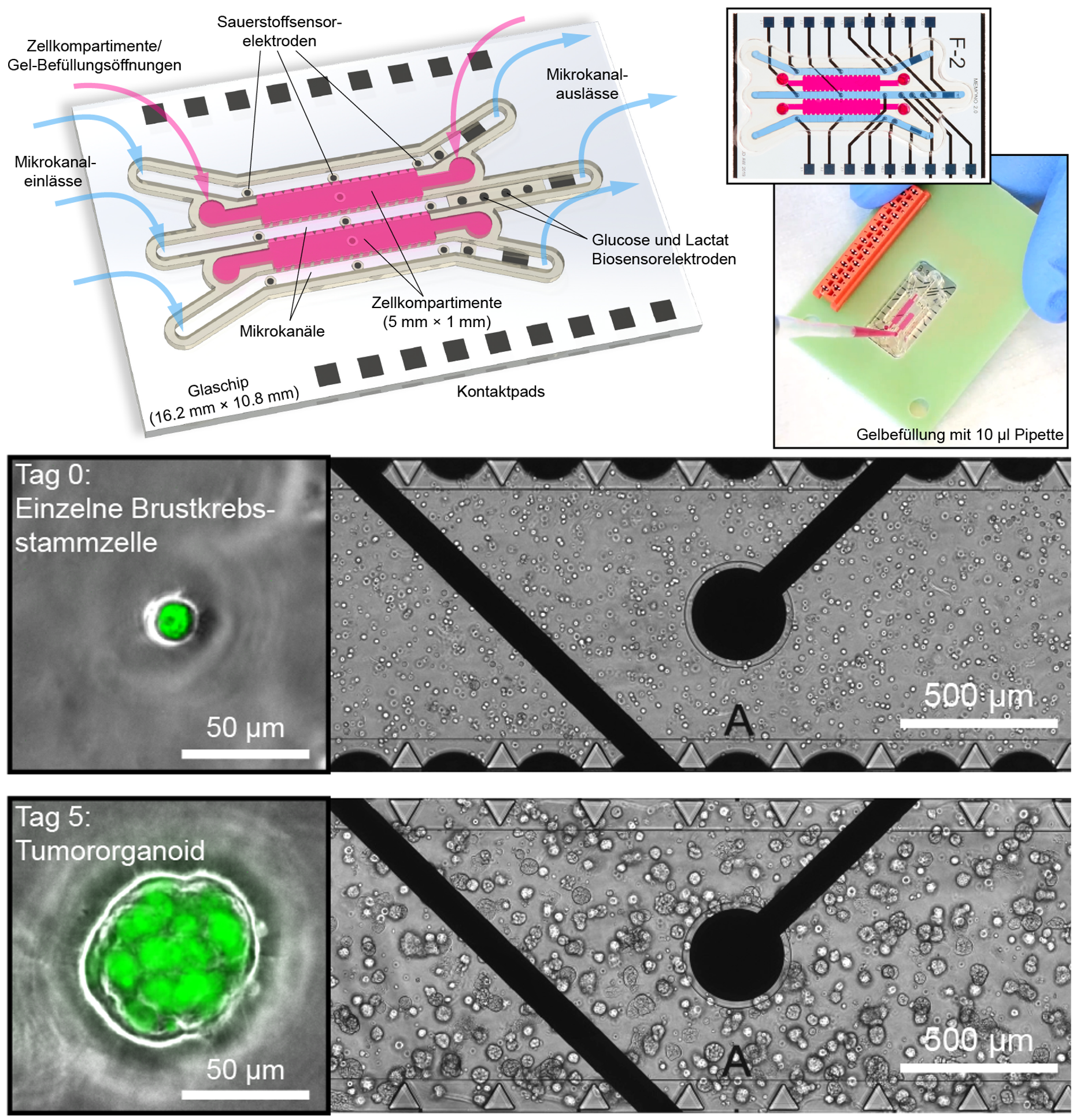 In organ-on-chip systems, stem cells are multiplied and examined in three-dimensional, organ-like assemblies. In addition to cell growth chambers (pink) and microchannels, the innovative variant from Freiburg also contains several different microsensors (black circles), which enable continuous monitoring. Below: small tumour organoids develop from individual breast cancer stem cells (green) within 5 days. © Andreas Weltin and Johannes Dornhof
In organ-on-chip systems, stem cells are multiplied and examined in three-dimensional, organ-like assemblies. In addition to cell growth chambers (pink) and microchannels, the innovative variant from Freiburg also contains several different microsensors (black circles), which enable continuous monitoring. Below: small tumour organoids develop from individual breast cancer stem cells (green) within 5 days. © Andreas Weltin and Johannes DornhofWith the help of the oxygen sensors, the researchers investigated the effect of the cancer drug doxorubicin using the stop/flow method as an example. After supply with fresh nutrient solution (flow phase), the oxygen contained in the solution decreases in the stop phase, as it is consumed by the living cells. The addition of the drug kills the cells, which can be seen by a reduced decrease in oxygen. Weltin reports enthusiastically: "Through the sensor signals, we notice immediately that the cells are no longer breathing, although they still appear healthy. This is a big advantage over conventional methods, where the result is sometimes only visible after a number of days." His team was also able to clearly show that the effective doxorubicin concentration determined in 2D cultures is not effective in organ-like cell assemblies, but that ten times the amount must be used here.
The organ-on-chip system also contains sensors for glucose and for the lactate (lactic acid) formed during oxygen deficiency. These parameters can be used to monitor the cell’s energy metabolism so that culture conditions can be optimally adjusted. The measurement of other parameters is also possible without any problems. "The sensor principles are the same everywhere. In a small electrochemical cell, the substance concentration is measured via a microelectrode," Weltin explains. "In each case, this is based on a platinum electrode, which is selectively converted into a specific sensor through modifications and corresponding measurement protocols."
Wide range of applications
An important advantage of real-time determination is the detection of temporary changes. Dornhof explains: "We can follow the cell metabolism live and thus also discover effects that disappear after some time. Such information is lost in endpoint analyses." For this reason, the novel system is particularly suitable for detailed studies on drug effects and could in future supplement or even replace animal experiments in some areas.
In the long term, an application in the field of personalised therapies is also planned. If the mini-tumours are grown from the patient's own stem cells, the effect of the drugs could be examined before treatment and the most suitable drug identified. A high degree of interdisciplinary cooperation between microsystems engineers, medical doctors and cell biologists would be required to make this happen.