Viral cancer therapy
Therapeutic viruses against tumours and metastases
Viruses can overcome cell barriers and transfer information to their host cells. They bring little material of their own to this, as they know how to make their host cell’s infrastructure work for them. This makes them excellent biotechnological tools, which a research group from the Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB in Stuttgart is using to its advantage. The team from the IGB’s Virus-based Technologies innovation field is developing a therapeutic virus that not only recognises and fights tumours, but also has the potential to reach metastases.
Many people have experienced it: it starts as a slight tingling sensation on the lip, then itchy blisters form. The causative agent of this annoying infectious disease is the herpes simplex virus type I (HSV1). It is widespread in the population, but only causes the symptoms described in 20 to 40 percent of those affected.1)
As it has evolved, the HSV virus has perfected the attack on the human host cell – and that is one reason why the research group led by Prof. Dr. Susanne Bailer at the Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB in Stuttgart is using it to develop new cancer therapies. The researchers in the Virus-based Technologies innovation field have succeeded in modifying the virus in such a way that it can recognise and even kill cancer cells while eliminating the pathogenic viral properties described above.
Strictly speaking, a virus is not a living organism, but a protein capsule in which a viral genome is enclosed. This contains the blueprint for the virus and its replication programme. Viruses recognise their host cells very specifically with the help of special receptors, dock onto the cells and introduce their genome into them. They multiply in the host with the help of the host’s cellular metabolism. The infected host organism weakens as the viral infection proceeds, resulting in the typical viral symptoms.
The potential of reprogrammed pathogens in cancer therapy
Viruses are excellent biotechnological tools thanks to their ability to cross cell boundaries and transmit information with a minimum of equipment. "The idea of reprogramming them into therapeutic viruses and using them in the fight against cancer is not new," explains Bailer. "But the coronavirus pandemic has given a huge boost to the use and further development of the technology." For example, Astra Zeneca's COVID-19 vaccine is based on the principle of a therapeutic virus: the genetic code for the vaccine antigen is introduced into human cells with the help of an adenovirus. Inside the human cell, the DNA is read and the antigen produced. This provokes the desired immune response. "The company's huge success with the world's first approved vaccine against COVID-19 has encouraged research into genetically modified viruses," says Bailer.
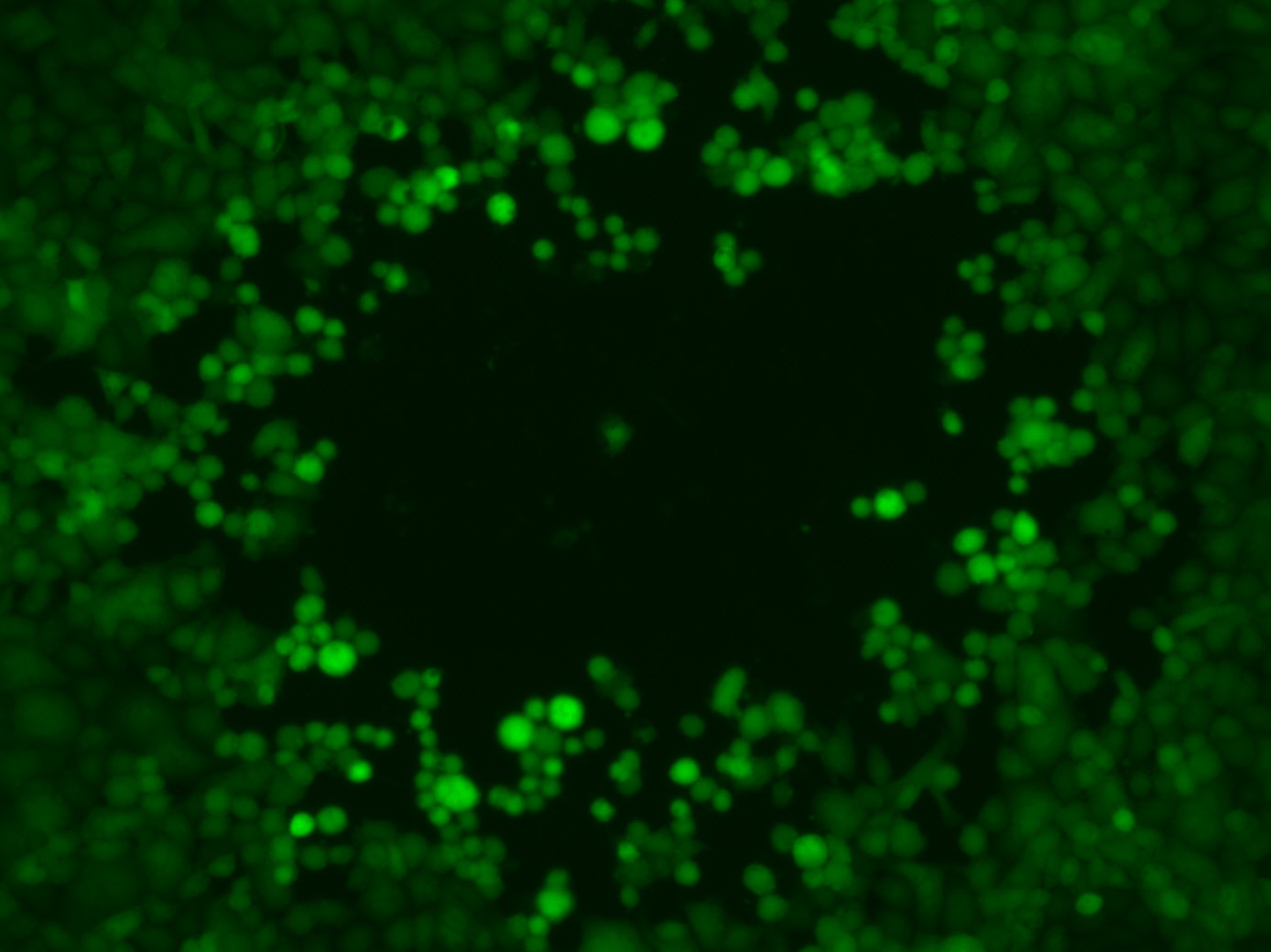 The oncolytic herpes simplex viruses (fluorescing green) multiply in the cancer cells, eventually causing them to burst. © Fraunhofer IGB
The oncolytic herpes simplex viruses (fluorescing green) multiply in the cancer cells, eventually causing them to burst. © Fraunhofer IGBA few years before the coronavirus pandemic, a therapeutic virus had already been approved for cancer therapy: talimogene laherparepvec (T-VEC) is a modified herpes virus developed for intratumoral injection and considered a pioneer for immunotherapy with oncolytic viruses and has been on the market since 2015 for treating malignant melanoma.2)
The virus exerts an oncolytic effected after injection into one or more of the patient's tumours. It multiplies in the tumour cells and eventually causes the cells to burst (Fig. 1). This leads to the release of certain tumour markers. These in turn put the immune system on alert and trigger a whole cascade of inflammatory and immune reactions that fight the cancer cells.
Unlike SARS-CoV-2 and other RNA viruses, HSV has a DNA genome. This means that the genetic information it carries is stored in the form of DNA inside the virus. "The HSV DNA genome is much larger than the genome of most viruses. Therefore, it can accommodate the many genes that are required for the desired function," says Bailer.
Reprogramming the virus requires two important steps: firstly, the viral genes that trigger the disease symptoms must be switched off. Secondly, and most importantly, the neurotropic properties must be suppressed, i.e., the ability of the herpes virus to migrate into the nerve tracts where they cause severe damage, such as meningitis.
Therapeutic viruses act locally and systemically
The functions, i.e., genes beneficial for fighting cancer, can be implanted into the virus that has been rendered harmless. This includes, for example, a feature that Bailer calls "targeting": once the viruses are injected into the tumour tissue, targeting means that they only attack cancer cells and not healthy cells.
In addition, other genes can also be inserted, for example those whose proteins trigger immunomodulatory effects in the patient's body. Among other things, it is possible to use the genetic code of so-called immune checkpoint inhibitors; these are antibodies that are already approved for cancer therapy today. The inhibitors dock onto certain checkpoints of the immune system and make cancer cells vulnerable to the body's own immune defence system. "The immune system is our strongest weapon against cancer. Therapeutic viruses, like the ones we have developed, can stimulate the immune system in a targeted way so that the body can better defend itself against degenerate cells," says Bailer. "The therapeutic virus therefore not only acts locally in the tumour, but also systemically (i.e. in the entire body) via the immune system. We are hoping that this will enable us to have an effect on metastases, should they arise."
TheraVision: development and test platform for therapeutic viruses in modular design
 Cell culture incubator © Fraunhofer IGB
Cell culture incubator © Fraunhofer IGBSimilar to the already approved T-VEC, the oncolytic herpes simplex virus developed by Bailer's working group is one day expected to be used as a vector for tumour immunotherapy. The researchers have applied for a patent for a modular platform technology to produce and test viruses for tumour therapy: with TheraVision, the individual therapeutic functions can be flexibly combined as needed, depending on the required target organ and mechanism of action.
The advantage of this modular engineering is that functionalised viruses can be produced and tested in preclinical models more quickly and easily than before. This speeds up preclinical development and facilitates the GMP (good manufacturing practise) process required for drugs. Bailer's team, together with several other Fraunhofer institutes, has already developed technologies for the viral therapy of non-small cell lung cancer (NSCLC) based on the TheraVision platform. Initial preclinical tests have been successful. Clinical tests for the combined virus-immunotherapy are in preparation.