 Dr. Michael Hirth, head of the pancreas research group at Mannheim University Hospital. © Private
Dr. Michael Hirth, head of the pancreas research group at Mannheim University Hospital. © Private"For patients suffering from pancreatic cancer who have experienced excruciating tumour-associated pain for many months, the reduction of pain would be the greatest thing - even greater than longer survival," reports Michael Hirth, who heads the pancreas research group at Mannheim University Hospital. The assistant physician continues: "More than 75 percent of pancreatic cancer patients experience tumour-associated pain, half of them so massively that existing pain therapies are not enough to ease the pain."
The pain experienced by patients with pancreatic cancer is mainly attributed to sensitised or damaged nerves. Until now, researchers had mainly suspected that the pain was caused by messenger substances released by the cancer cells. "It was thought that the tumour’s messenger substances make nerves more sensitive, leading to cancer patients experiencing stimuli that would normally not trigger such bad pain," Hirth explains. Nine years ago, Hirth also showed in his doctoral thesis that a certain messenger substance, which is released by tumour cells among others, can sensitise nerve cells.
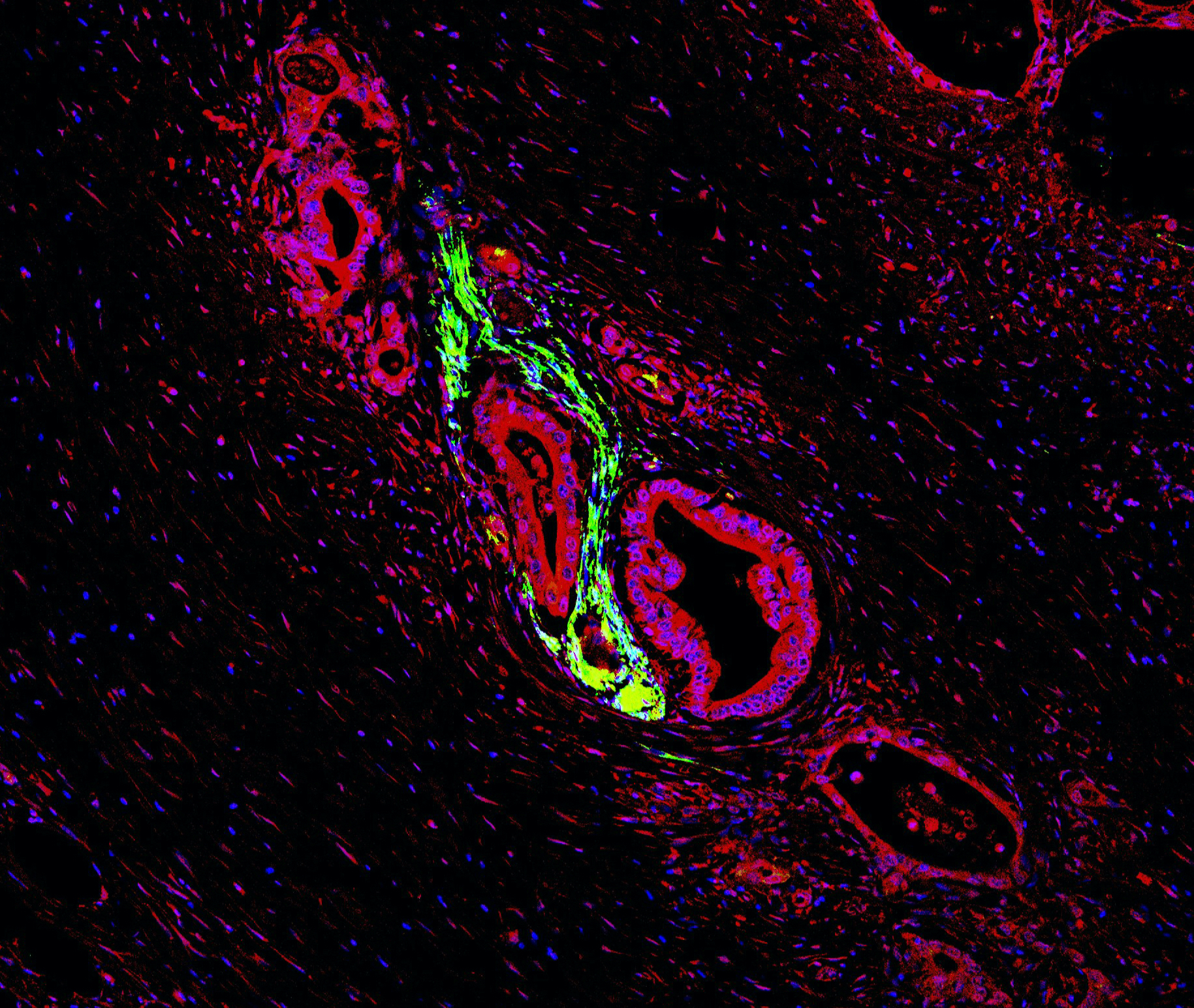 Tumour cells with high expression of a chemokine receptor (red) have infiltrated the nerve (green) and are beginning to damage it. The tissue comes from a patient suffering from pancreatic carcinoma and tumour-associated pain. Blue dots mark cell nuclei. © Institute of Pharmacology, University of Heidelberg
Tumour cells with high expression of a chemokine receptor (red) have infiltrated the nerve (green) and are beginning to damage it. The tissue comes from a patient suffering from pancreatic carcinoma and tumour-associated pain. Blue dots mark cell nuclei. © Institute of Pharmacology, University of HeidelbergHowever, this does not explain why pancreatic cancer cells seem to be magically attracted to nerve cells and are preferentially found in their vicinity. As the Mannheim researcher, together with scientists led by the renowned pain researcher Prof. Dr. Rohini Kuner from the University of Heidelberg, found out, nerve cells themselves secrete attractants.1 "Presumably, nerve cells need these messengers for self-stimulation in order to survive," says Hirth.
However, the researchers found that the chemokines CXCL10 and CCL21 in particular had the disastrous effect of attracting pancreatic cancer cells that were characterised by a significant upregulation of the relevant surface receptors. When the scientists blocked one of the two chemokines or their receptors in mice with pancreatic cancer, the animals experienced much less pain. This finding was substantiated by the fact that the researchers found a greater number of CXCL10- and CCL21-relevant receptors on pancreatic cancer cells in patient biopsies. "Patients with a high receptor density on the tumour cells also frequently experienced tumour-associated pain," reports Hirth.
Award-winning research
The physician won first prize for his research in the German Pain Society’s Basic Research awards in October 2020. In future, Hirth would like to use the findings from these studies to identify patients with pancreatic cancer whose tumours have an excessive number of these two receptors. Such patients could particularly benefit from early pain therapy.
"In the long term, the idea would be to treat these patients with chemokine-blocking substances, but that is still a pipe dream," says Hirth. The problem is that chemokines normally also attract immune cells, which is actually desirable in the fight against cancer cells. "First of all, we have to carry out preclinical studies to identify which chemokines we can inhibit without suppressing the immune response," says Hirth.
Communication between pancreatic cancer cells and nerve cells is very complex, and the researchers believe that other nerve messenger substances yet to be investigated are also involved. "We also suspect that the tumour cells release mediators which trigger the overexpression of messenger substances in the nerve cells," notes Hirth. The fact that cancer cells migrate to and invade nerve cells has several advantages for the tumour. Hirth explains why: "The neuronal compartment is very rich in nutrients, quite well protected from the immune system, and the tumour cells can metastasise very effectively along nerves," explains Hirth.
One of the most painful tumour types
The affinity of tumour cells for nerves is probably one of the reasons why patients with pancreatic cancer are more susceptible to tumour-associated pain than with other forms of cancer. In addition, the pancreas is in relatively close vicinity to pain-processing nerve clusters along the spinal cord that transmit pain stimuli to the brain.
If pain caused by pancreatic carcinoma or a chronically inflamed pancreas persists for years, there is a risk that patients will develop a pain memory. "Here, the main event is no longer the activation of pain receptors in the pancreas, but sensitisation in the brain," says Hirth.
Hirth's research group is currently developing a hypersensitisation test to identify patients who have already developed a pain memory. The researchers are using the test to detect the threshold at which such patients perceive stimuli such as heat or cold in different parts of the body as painful. "Our hypothesis is that those patients who have not yet developed a pain memory need a different pain therapy than those who have developed one," says Hirth, who has been fascinated by the topic of pain ever since he attended a lecture on the subject while he was at medical school. In future, thanks to personalised pain therapies, tumour-associated pain could at least become more bearable for patients with pancreatic cancer, even if prognosis would remain very poor.