Fatal, incurable and often diagnosed far too late - pancreatic cancer is on track to become the second leading cause of cancer-related deaths, driven by a continuously rising incidence. What makes it such an extraordinary challenge for medicine?
The course taken by this particular disease is remarkably heterogeneous: in many ways, every patient has a distinct form of pancreatic cancer, which makes comparisons and standardised treatment approaches difficult. The reasons for these huge differences are only partly understood. Much depends on the tumour’s molecular profile, but the surrounding tumour microenvironment also plays a crucial role. In pancreatic cancer, the immune system is effectively shut out. The tumour releases numerous factors that suppress immune activity, preventing the body’s natural defenses - and even modern immunotherapies - from mounting an effective response.
 Prof. Dr. Thomas Seufferlein, Medical Director of the Clinic for Internal Medicine I at University Hospital Ulm, leads the UNITEPANC trial. © University Hospital Ulm
Prof. Dr. Thomas Seufferlein, Medical Director of the Clinic for Internal Medicine I at University Hospital Ulm, leads the UNITEPANC trial. © University Hospital UlmYou have been working as a physician and researcher in this field for more than two decades. Has there been progress in the diagnosis and treatment of this disease?
We have significantly extended survival times. In 2000, the average survival time was around two years; today, after surgery, it is about 54 months. However, we have made little progress in early detection. In patients with metastatic disease, survival is still only eleven to twelve months. We are now seeing the emergence of a new class of drugs: RAS inhibitors [editor’s note: around 80 percent of tumours harbour RAS mutations].
Effective inhibitors that target mutated RAS have been developed over the past three to four years. These agents could fundamentally change the therapeutic landscape for pancreatic cancer. A large trial in which we were also involved (NCT06625320) has just been completed, and the results are eagerly awaited. For the first time, we may be able to directly target one of the tumour’s key mutations.
What are these new drugs?
They are RAS inhibitors that either selectively target specific mutations in RAS proteins or, in the case of pan-RAS inhibitors, act more broadly. Depending on the approach, they differ in efficacy and side-effect profiles. Pan-RAS inhibitors can suppress mutated RAS as well as the endogenous RAS protein, which increases the risk of side effects - for example, because stem cells in the gut also rely on RAS signalling for correct differentiation.
What are UNITEPANC’s objectives?
UNITEPANC has two major objectives. The first is to improve adjuvant chemotherapy after pancreatic cancer surgery. We do this by creating miniature versions of patients’ tumours - called organoids - from the tissue removed during surgery. These organoids are then exposed to different chemotherapy drugs to determine which treatments the tumour is most sensitive or resistant to, thereby tailoring therapy to the individual patient.
The second objective of our Phase I trial is to show that this approach can be reliably reproduced across multiple clinical centres and is robust enough to be implemented in routine clinical practice.
Why is this so difficult?
The process is highly complex and prone to errors. We conducted round-robin studies across multiple sites over a two-year period. The current challenge is to determine whether this approach is feasible in a real-world clinical setting. The key question is whether organoid generation can be decentralised across sites, or whether it must be centralised to ensure reliability if it is to be implemented in clinical practice.
How can the benefits for patients be described?
If organoid testing shows that the same therapeutic effect could be achieved with a less aggressive regimen than the maximum standard therapy, we can significantly reduce treatment-related toxicity. This means patients would undergo therapy that is both effective and far less physically and mentally taxing.
Is UNITEPANC the world’s first multicentre study?
The superlative refers to the fact that, to date, this approach has not been evaluated in a multicentre, prospective, randomised controlled trial. Most existing studies follow the reverse paradigm, whereby organoids are generated within the context of an ongoing clinical study. In contrast, a multicentre study designed to generate results that directly inform clinical decision-making has not yet been undertaken, largely due to its inherent complexity. We aim to provide a proof-of-concept by addressing the critical question of whether this technology can be successfully translated into clinical practice.
What proportion of patients are eligible for surgery?
No more than 20 percent.
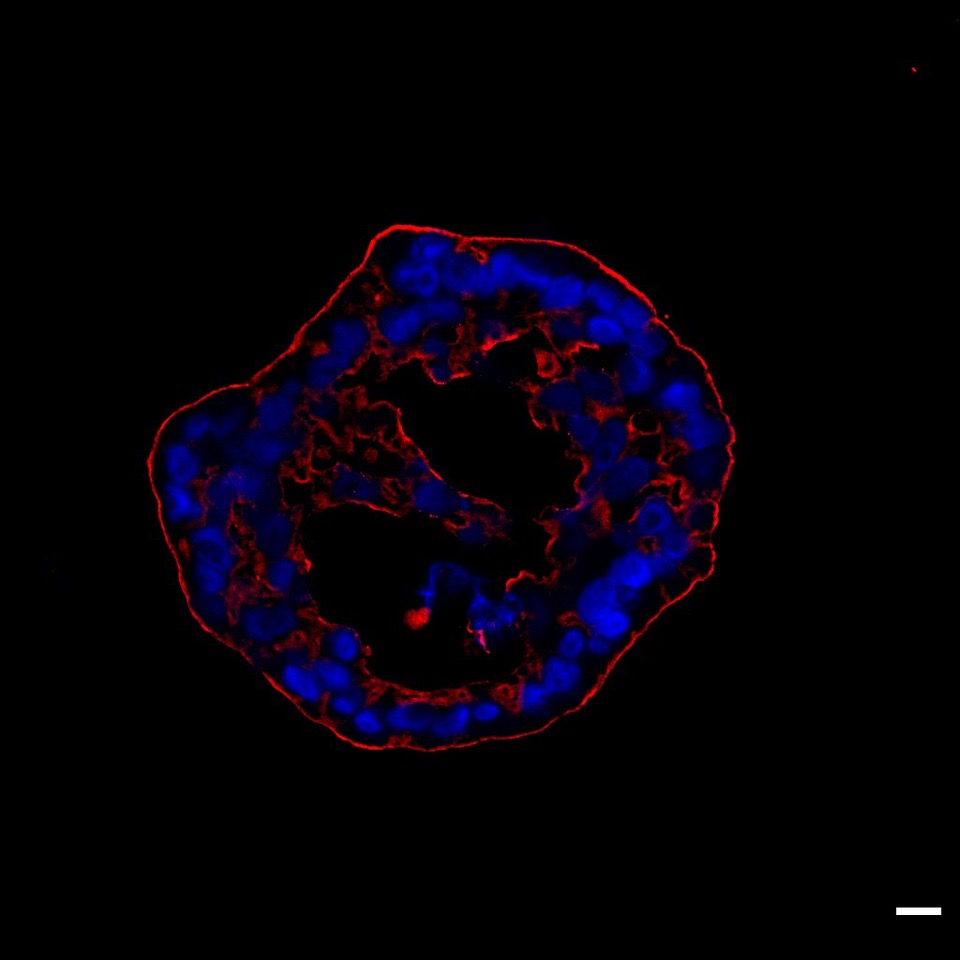 Will organoids make it into routine clinical practice? Our image shows a patient-derived immunofluorescent organoid. © IMOS, Prof. Alexander Kleger, University Hospital Ulm
Will organoids make it into routine clinical practice? Our image shows a patient-derived immunofluorescent organoid. © IMOS, Prof. Alexander Kleger, University Hospital UlmThat is, in essence, a modest goal...
That is one of the key problems with this disease: we lack diagnostic tools for early detection. Pancreatic cancer is a tumour that releases very little tumour material into the blood and is difficult to detect in the blood in its early stages. All the symptoms are non-specific. Many older patients (in their 60s to 80s) are treated for back pain until it turns out that a pancreatic tumour is growing along the nerve sheath. The pain is referred to the back, but actually originates from a pancreatic tumour. Even very good imaging is not yet perfect and can only detect early-stage tumours to a very limited extent and in isolated casesAuch sehr gute Bildgebung ist noch nicht perfekt und kann Frühformen nur sehr bedingt und in Einzelfällen erkennen.
It sounds as though early diagnosis remains difficult.
It absolutely is difficult and a major challenge for research. Here too, RAS mutations play a role, representing early changes in the development of pancreatic carcinomas. The problem is simply this: these changes may, but do not necessarily, lead to a tumour. Autopsy studies show that quite a few people have precancerous lesions in the pancreas but do not develop cancer..
Are there any existing organoid technologies that can be used to produce tumour-based organoids anywhere using the same method, which would make them comparable and reproducible?
There are indeed attempts by companies to standardise this process. However, I have not yet seen any prospective trials from them to validate their approach. At this stage, they are still largely in the research phase.
Does the intratumoral heterogeneity of a primary tumour persist in these organoid ‘avatars’ in the same way as it does in vivo?
Organoids lack the full tumour microenvironment - such as tumour-associated connective tissue cells, macrophages, collagen and stroma. Nevertheless, our retrospective studies have shown that the correlation between organoid responses and patient outcomes are surprisingly good. In another project, we are attempting to address this limitation using a 3D printer to ‘print’ tumour cells together with matrix components, macrophages and immune cells. It remains to be seen whether this approach will allow us to predict tumour responses more accurately. While this works well with established model cell lines, it is more challenging with patient-derived cells because they survive outside the body to varying degrees.
How long can organoids be propagated for while preserving clonal heterogeneity?
To avoid selecting for specific clones during cell culture, we aim to use low passage numbers. This is not a limitation for our testing approach, because we establish the cultures and test them immediately. By using the lowest possible passages, we preserve the original heterogeneity of the tumour.
A universal SOP for organoids assumes that it does not matter when or under what conditions a biopsy is taken.
In reality, it does matter: the timing of the biopsy can have a significant impact. Whether an organoid can be successfully established from a tumour sample depends, among other factors, on whether and how many cycles of chemotherapy the patient has already received. The more prior treatments, the more difficult it becomes to generate organoids. Interestingly, organoids can often be obtained more easily from metastases than from the primary tumour. There are still aspects of the technology that we do not fully understand.
Given the complexity of the procedure, is it realistic to bring this into clinical practice?
Is it feasible? I believe so. Achieving this in practice is realistic. The critical question is how often we succeed - 90, 100 or 50 percent of the time. Our patients always receive the best possible treatment, even if we are unable to establish an organoid. Once sufficient data have been accumulated and can be used to train predictive models, we may be able to define predictors that will allow direct testing on tumour material in the future - potentially bypassing the ‘detour’ via organoids.