"We are more than the sum of our genes" is how epigeneticists describe their field of research. Genetic information stored in the DNA does not just determine our appearance and behaviour, but also when and where this information is read. Epigenetics, from the Ancient Greek prefix ‘epi’ meaning ‘on top of ’or ‘in addition to’, and the term ‘genetics’, describes factors beyond the genetic code. Epigenetic DNA modifications regulate when individual genes are turned on. Epigenetic changes in the form of chemical modifications to or near the DNA thus create conditions that enable or prevent gene transcription. As we develop from a fertilised egg cell to an organism, specific epigenetic modifications occur in specific cells at certain points in time, enabling cells to be differentiated into different cell types (e.g. heart, liver, skin cells) despite the fact that all our body’s cells contain the same genetic information. "Once the decision [in one direction or another] has been made, it is remembered and fatefully propagated," explains Dr. Asifa Akhtar, Director at the Max Planck Institute of Immunobiology and Epigenetics in Freiburg.
Sex chromosome dosage compensation is key to survival
All genes are subject to epigenetic regulation. DNA methylation is the longest known epigenetic mechanism that modifies gene function. DNA methylation involves adding a small chemical compound (methyl group; -CH3) to a DNA building block (predominantly cytosine bases). If this modification takes place within regulatory sequences, transcription of the associated gene is inhibited. This mechanism plays an important role in X-chromosome inactivation in mammals, among other things. Female individuals have two X chromosomes in their cells, whereas male cells have one X and one Y chromosome. However, a defined level of X chromosome gene products is required for the organism to develop normally, which means that the genetic inequality between the two genders must be corrected. This is achieved by the epigenetic inactivation of one of the two X chromosomes in female cells, a process also known as dosage compensation.
It is interesting to note that the model organism used in developmental biology, the fruit fly Drosophila melanogaster, solves this problem differently. Dosage compensation in D. melanogaster is achieved by the twofold transcriptional upregulation of the X chromosome in males, so that the resulting amount of protein corresponds to the amount of protein in female cells with two X chromosomes.
MOF histone acetyl transferase facilitates gene transcription
In all eukaryotes (living organisms with a cell nucleus), the filamentous DNA strands are compacted into chromosomes in order to fit into the cell nucleus. The smallest packaging units are called nucleosomes, which consist of special proteins called histones around which a segment of DNA is wrapped. The nucleosomes form what is known as beads-on-a-string structures, which in turn coil into chromatin superstructures (consisting of proteins and DNA), a compact structure which suppresses gene transcription. The properties of the histones determine how tightly the DNA is bound and how close together the nucleosomes are located. The compact (condensed) chromatin state results from the strong attraction between the positively charged histones and the negatively charged DNA.
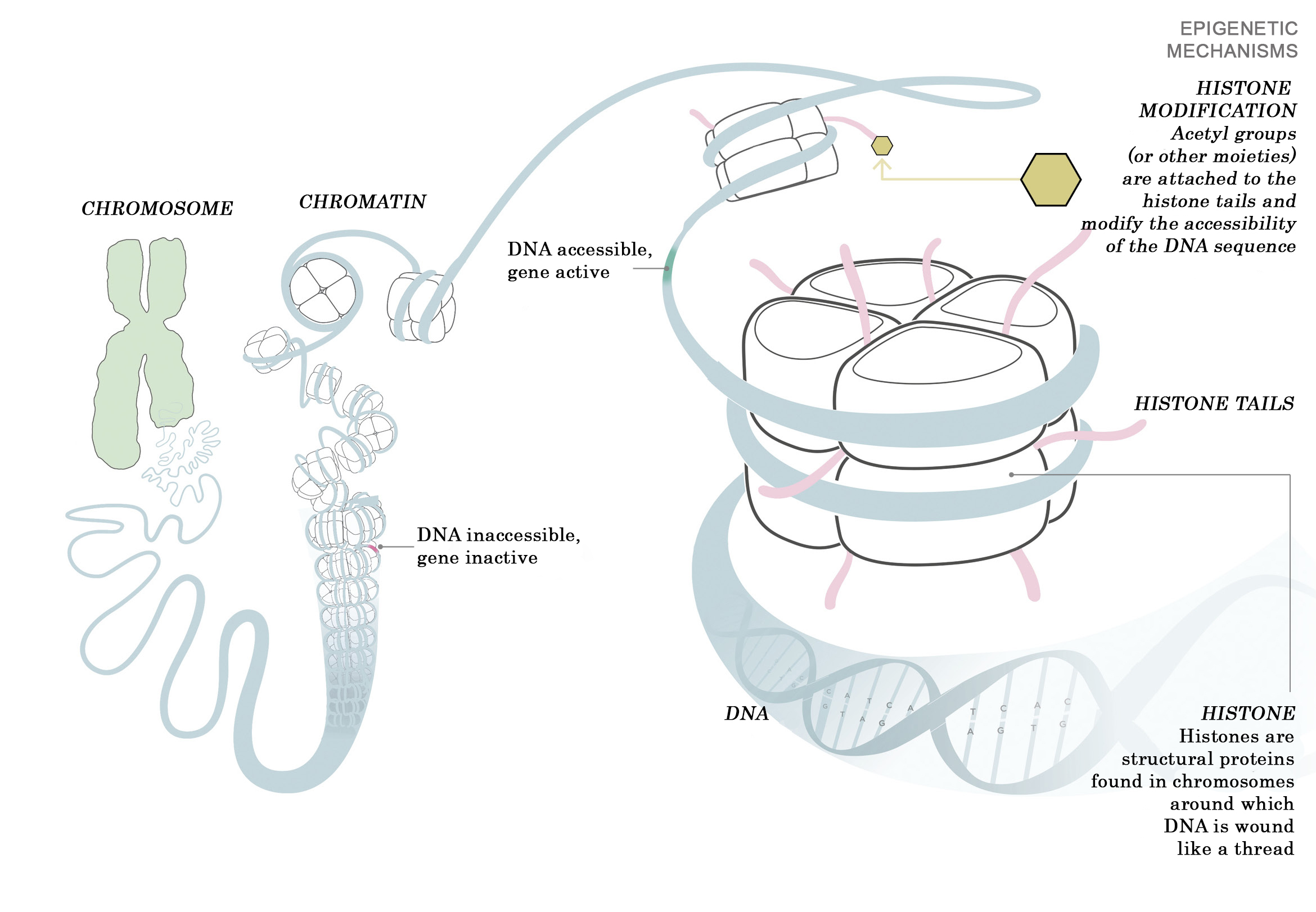 Schematic representation of chromatin and chromosome organisation and histone modifications. © GCO modified from MPI of Immunobiology and Epigenetics drawing
Schematic representation of chromatin and chromosome organisation and histone modifications. © GCO modified from MPI of Immunobiology and Epigenetics drawingIn Drosophila, the MSL (male-specific lethal) complex, which consists of at least 5 proteins (MOF, MSL1, MSL2, MSL3, MLE) and two non-coding RNAs (roX1 and roX2), is responsible for activating the X chromosome genes. MOF (males-absent on the first protein) is a catalytically active protein (enzyme), a histone acetyl transferase, which replaces the positive charge of a histone with a neutral acetyl residue (-COCH3). This weakens the contact between histone and DNA and loosens (opens) the affected region, making it accessible to the transcription machinery.
Asifa Akhtar has been studying the phenomenon of dosage compensation in Drosophila melanogaster and the MOF enzyme for twenty years. "My area of expertise is chromatin biology. The way you open and close chromatin, how you can make different combinations of accessible genes is the key to epigenetic regulation," she explains. Her work has elucidated many fundamental mechanisms, work for which she was awarded the Leibniz Prize by the German Research Foundation (DFG) in March 2021. Recent research carried out by her research group illustrates how the MSL complex can distinguish the X chromosome from autosomes (non-sex chromosomes) and why the loss of MOF in mammals leads to nuclear instability and chromosomal abnormalities, as is often the case in cancer cells.
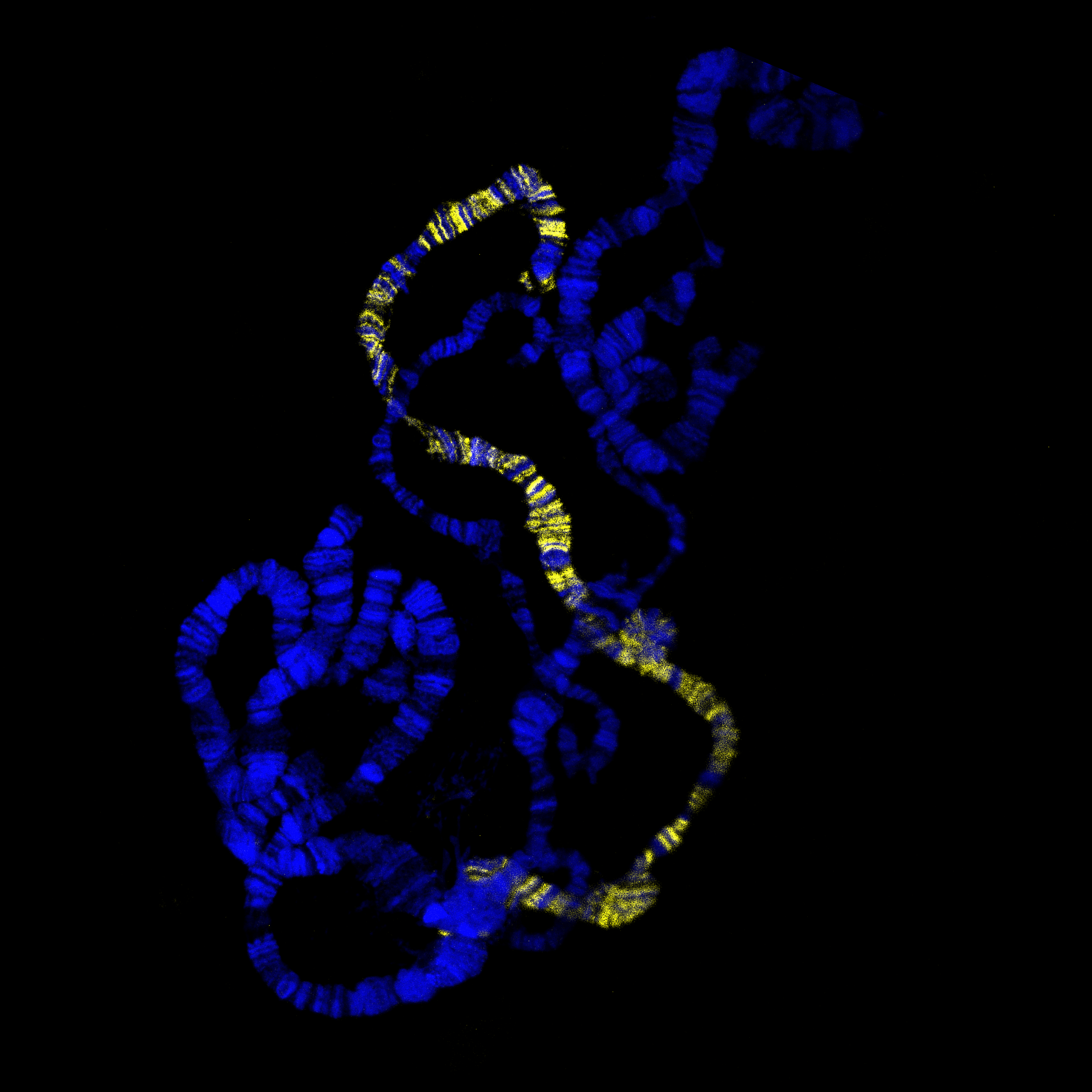 Chromosomes of a male Drosophila fly. The yellow stained MSL complex binds to the X chromosome, the other chromosomes are stained blue. © Aline Gaub, MPI of Immunobiology and Epigenetics.
Chromosomes of a male Drosophila fly. The yellow stained MSL complex binds to the X chromosome, the other chromosomes are stained blue. © Aline Gaub, MPI of Immunobiology and Epigenetics.MOF regulates transcription in nucleus and mitochondria
As far back as a decade ago, Akhtar's research group discovered that the Drosophila melanogaster MOF enzyme is also a component of the NSL (non-specific lethal) complex, which is involved in the regulation of genes on the autosomes. Both complexes are evolutionarily conserved, meaning they also occur in very similar forms in mammals. In studies on human cells, Akhtar's team was able to show that the NSL complex not only regulates genes in the cell nucleus, but also in the mitochondria. The latter are membrane-enclosed structures in the cell that produce energy and have their own DNA. "This leads us to another level of biology we can now address: The importance of communication between nucleus and cytoplasm and between organelles," the researcher explains. The mammalian NSL complex appears to be a metabolic sensor and to coordinate gene transcription in the nucleus and mitochondria.
 Dr. Asifa Akhtar, Director at the Max Planck Institute of Immunobiology and Epigenetics in Freiburg, Germany, studies the multiple functions of the MOF enzyme. © Marcus Rockoff, MPI of Immunobiology and Epigenetics
Dr. Asifa Akhtar, Director at the Max Planck Institute of Immunobiology and Epigenetics in Freiburg, Germany, studies the multiple functions of the MOF enzyme. © Marcus Rockoff, MPI of Immunobiology and EpigeneticsTo better understand how the proteins in the MSL complex function and interact, Akhtar's team carried out structural analyses, among other things, to show which parts of the proteins interact with each other. These findings took on new significance a few years ago when the scientist was contacted by doctors in France who had identified mutations in the MSL3 protein in children with a rare neurological syndrome. Based on the available data, it became apparent that the changes in MSL3 prevented the binding of the protein to the histone acetyl transferase MOF. The researchers found that this severely limits the enzymatic activity of MOF, so that only a reduced number of essential epigenetic marks can be set. This causes developmental delays and progressive neurological disorders. "Although it’s a genetic mutation, it mirrors the epigenetic alteration of the chromatin," says Akhtar. This opens up therapeutic possibilities because epigenetic modifications can be influenced and changed. In cell culture experiments, the researchers were able to show that so-called histone deacetylase inhibitors, which stabilise the acetylation of histones and are used in cancer therapy, lead to an improvement. These results provide important starting points for initial treatment options.
The decoding of the disease now known as Basilicata-Akhtar syndrome demonstrates the importance of basic research. The Max Planck Institute offers fantastic conditions for carrying out basic research and also gives researchers stability. "We can address a question in depth. I am very grateful. […] Investing in proper science is very important," explains Akhtar, who has been Vice President of the Max Planck Society since summer 2020. The additional support from the DFG within the MEDEP Collaborative Research Centre and the CIBSS (Centre for Integrative Biological Signalling Studies) cluster of excellence also enables productive collaborations with many departments at the University of Freiburg.