Nanoparticles as drug carriers
Inhalation of nanocarriers for antibiotics against resistant tuberculosis pathogens
Around ten million people worldwide still contract tuberculosis (TB) every year. With an estimated 1.4 million deaths a year, tuberculosis was the world’s deadliest infectious disease until COVID-19. The high mortality rate is down to the sophisticated biology of the pathogen Mycobacterium tuberculosis. As part of a collaborative BMBF-funded project called ANTI-TB, a team of researchers from the Karlsruhe Institute of Technology (KIT) and the Research Centre Borstel - Leibniz Lung Centre (FZB) has developed a method that aims to outsmart the bacterium once and for all. The method involves nanoparticles developed by Prof. Dr. Claus Feldmann at KIT, which were then successfully tested by Prof. Dr. Ulrich Schaible at the FZB in the form of an aerosol for their effectiveness against the tuberculosis pathogen.
The "tubercle bacillus" was first discovered and described by Robert Koch in 1882, for which he received the Nobel Prize in Physiology or Medicine in 1905. Nowadays, the disease is mainly treated with antibiotics. Although a TB vaccine is available, the German Standing Committee on Vaccination (STIKO) stopped recommending it in 1998 after it was found that the vaccine is not sufficiently effective against pulmonary TB in adults.
Overall, there is a worrying increase in tuberculosis cases caused by antibiotic-resistant pathogens, particularly as a result of poverty and lack of effective healthcare measures in poorer countries. Even in Germany, three percent of the approximately 4,000 TB cases a year are caused by multidrug-resistant TB bacteria. In Germany, all TB cases have to be reported to the Robert Koch Institute. Due to the COVID pandemic, progress in controlling TB has slowed or even reversed.
Disease remains largely undetected
"It is assumed that about a quarter of the world's population is infected with tuberculosis," says chemist Prof. Dr. Claus Feldmann, who studies nanoparticles at KIT. "The disease is very infectious: people with TB can spread it to others they are in contact with." Tuberculosis can affect many organs, but around 90 percent of cases involve the lungs. Infection usually occurs by inhaling aerosols containing bacteria that are coughed up by infected individuals. However, only five to ten percent of those infected actually become ill during their lifetime; in the remaining 95 to 90 percent, tuberculosis usually remains clinically inconspicuous (latent) and is not contagious.
The pathogens are often fought off by the immune system as they enter the respiratory tract. However, if the immune system does not recognise them, they can settle in the lungs where they become encapsulated and remain in the body for years without causing symptoms. When a latent TB infection is reactivated, the pathogens multiply again massively and the exuberant inflammatory response causes granulomas to burst open. This results in open tuberculosis, which causes fatigue, fever and coughing. In severe cases, lung tissue is destroyed, and coughed up along with blood in the sputum, leaving holes in the lungs. The body then puts all its energy into fighting the infection. The term "consumption", once coined by Hippocrates, describes how sick people become weaker and weaker, in effect, wasting away. If left untreated, TB can be fatal.
Clever mycobacterium
 Prof. Dr. Claus Feldmann at the Karlsruhe Institute of Technology (KIT) is developing nanocarriers, the smallest transport containers that deliver antibiotics to bacteria. © Prof. Dr. Claus Feldmann, private
Prof. Dr. Claus Feldmann at the Karlsruhe Institute of Technology (KIT) is developing nanocarriers, the smallest transport containers that deliver antibiotics to bacteria. © Prof. Dr. Claus Feldmann, privateThe main treatment method for TB consists of a combination of four different antibiotics, which a patient has to take for the first two months of treatment, followed by two of the antibiotics for a further four months. Resistant strains of the pathogen can jeopardise a successful outcome and are increasingly frequent when antibiotic treatment is discontinued too early. The World Health Organization (WHO) therefore recommends that antibiotic treatment be continued for at least six months after symptoms have disappeared. However, these oral antibiotics can lead to unwanted side effects including hearing loss and liver and kidney damage. This in turn reduces patient compliance, especially when people start to feel better. "If treatment is discontinued too early, there is a huge risk that some of the bacteria will survive and develop resistance to the antibiotics because the concentration of active substances at the site of action is no longer high enough," Feldmann explains. Treatment can take up to two years.
The rod-shaped pathogen Mycobacterium tuberculosis also uses clever strategies to evade the immune system. Normally, phagocytes engulf invaders in the lungs in a process known as phagocytosis and then eliminate them. However, Mycobacterium tuberculosis bacteria contained in phagosomes have glycolipids in their cell wall that interfere with the phagocytic digestion of the bacteria, thus ensuring their own survival within the phagocytes. The immune system responds by forming a wall of different defence cells around the site of inflammation. This leads locally to tissue destruction (necrosis) and the formation of a tuberculous granuloma, which isolates the pathogen in situ and prevents further spread. In response to encapsulation, the mycobacteria alter their metabolism, using fats from the dead tissue as a carbon source instead. The pathogens can also temporarily halt their metabolism altogether and remain in a state of dormancy. This further slows or even stops their growth.
Nanobased drug carriers
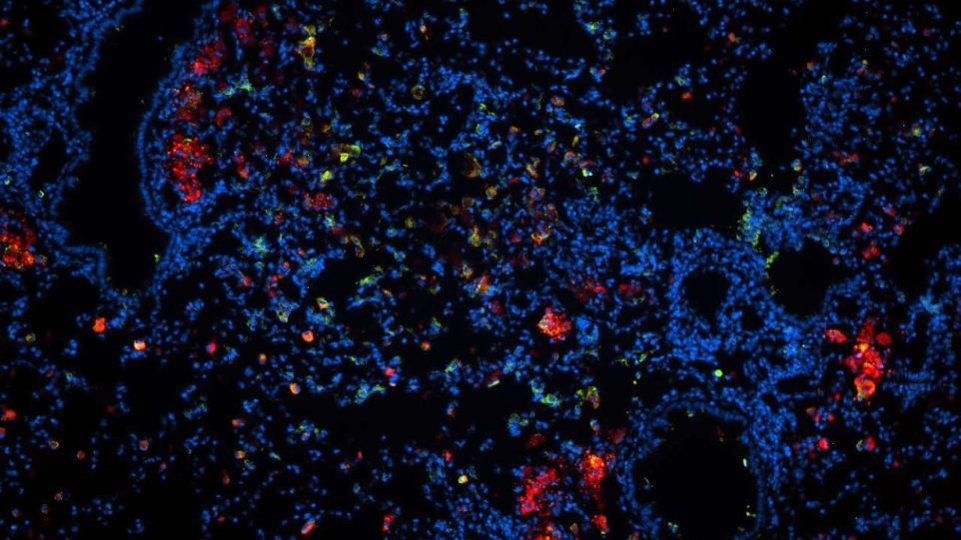 Infected lung tissue with cell nuclei (blue) and Mycobacterium tuberculosis (green) in which the antibiotic nanocarriers (red) have arrived at the site of infection. © Dr. Natalja Redinger, Research Centre Borstel - Leibniz Lung Centre (FZB)
Infected lung tissue with cell nuclei (blue) and Mycobacterium tuberculosis (green) in which the antibiotic nanocarriers (red) have arrived at the site of infection. © Dr. Natalja Redinger, Research Centre Borstel - Leibniz Lung Centre (FZB)Antibiotics that interfere with cellular processes are ineffective in treating Mycobacterium tuberculosis infections. The high mutation rate of the bacteria, even when dormant, makes the hunt for the pathogens even more difficult. A single mutation at a critical point in an enzyme can be enough to eliminate the point of attack for drugs. Mycobacterium tuberculosis also has an efflux pump that gives it the ability to selectively transport substances out of the cell, rendering most antibiotics ineffective. In addition to multidrug-resistant bacteria (resistant to at least two common antibiotics), extremely resistant bacteria also have a tolerance to fluoroquinolones. The latter account for about ten percent of antibiotic resistance worldwide.
"In addition to finding new active substances, we also need to find new methods of administration that ensure the active substances reach the site of infection in sufficiently high concentrations," says Feldmann. The problem is that most antibiotics are lipophilic, which means that they cannot be transported in aqueous environments such as the lung surface. Feldmann and his team have now produced antibiotic-loaded nanocarriers that can be effectively delivered to the target site. The researchers’ idea is to increase the local concentration of antibiotics in order to reduce treatment time and side effects. Patients might then be more willing to persevere with treatment. "Since TB is primarily a lung disease, our approach was to develop a method to inhale very small particles as an aerosol directly through the lungs," explains the chemist. "What makes our approach unique is that the nanocarriers are very small, only 30 nanometres, and that the small nanoparticles consist almost entirely of an antibiotic." The researchers pack the nanoparticles with high doses of new antibiotics: bedaquiline (BDQ) inhibits the enzyme ATPase in dividing and non-dividing bacteria, benzothiazine (BDZ-043) inhibits a bacterial enzyme that is needed to build the cell wall and causes the cell to dissolve.
Administered in the same way as an asthma spray
 Prof. Dr Ulrich Schaible at the Research Centre Borstel - Leibniz Lung Centre (FZB) is in charge of the medical side of the project and for testing its feasibility in living systems. © Prof. Dr. Ulrich E. Schaible, private
Prof. Dr Ulrich Schaible at the Research Centre Borstel - Leibniz Lung Centre (FZB) is in charge of the medical side of the project and for testing its feasibility in living systems. © Prof. Dr. Ulrich E. Schaible, privateHowever, these two active substances are also fat-soluble (lipophilic) and need to be dispersed when transported through an aqueous environment. After synthesis, the nanoparticles have a high drug load of 70% BDQ and even 99% BTZ-043 of the total nanoparticle load, plus a certain amount of surfactant (31% for BDQ, 1% for BTZ), which makes the lipophilic antibiotics water-dispersible. The researchers use a chemical trick to make the lipophilic drugs water-dispersible: the fat-soluble antibiotic/surfactant solution is quickly injected into water. The surfactant is soluble in water while the antibiotic precipitates immediately after injection and combines with the surfactant contained in the water. "This is how we produce the nanoparticles; we can also produce dispersions that remain stable for several weeks," says Feldmann.
Researchers at the Research Centre Borstel have shown that the infected cells take up the nanoparticles very well and that the active substance actually reaches the bacteria. One of the researchers, Prof. Dr. Ulrich Schaible, who studies host-pathogen interactions in tuberculosis and tested the biodistribution and efficiency of the nanocarriers in tuberculosis-infected mice, was convinced by the results that the method works, i.e. that the antibiotic nanoparticles are indeed delivered in high concentrations to the tuberculosis foci in the lungs, where they act against intracellular mycobacteria. Studies in infected mice showed that the innovative nanocarriers were up to 50 percent more effective than conventional pulmonary inhalers. By administering the nanoparticles directly into the lungs, side effects can be minimised and bacterial resistance is expected to be reduced. Two antibiotics with different modes of action can probably be combined in one carrier and inhaled as a liquid or dry aerosol spray. Nevertheless, further studies will take at least another five years before this promising therapy will be seen in everyday clinical practice.