Photoimmunotherapy
Switching off tumours with light: how photons can be used to target prostate cancer
Prostate cancer is the most common cancer among men in Germany. Relapses and the harsh side effects of traditional treatments remain a major challenge, even with advanced surgical techniques such as da Vinci robot-assisted surgery. At the University Medical Centre Freiburg’s Department of Urology, Dr. Isis Wolf is pioneering the development of photoimmunotherapy - a cutting-edge approach that could become a molecular milestone in cancer research. Photoimmunotherapy combines light, antibodies and a specialised dye to precisely target tumour cells while simultaneously 'waking up' the immune system.
Prostate cancer is tricky because it often has no symptoms in its early stages when the chances of a cure are highest. By the time urinary issues or bone pain manifest, the tumour has often already spread. The standard treatment, radical prostatectomy (complete removal of the prostate), still leads to a recurrence of the cancer in up to 40 percent of cases. Patients also face serious side effects such as incontinence or impotence, since delicate nerves run through the surgery site. At the University Medical Centre Freiburg, Dr. Isis Wolf and her Antibody-Based Diagnostics and Therapy research team are developing a gentler, molecular-based approach to complement surgery and reduce the risk of recurrence.
A molecular navigation system
 Biologist Dr. Isis Wolf focuses on prostate cancer at the University Medical Centre Freiburg, aiming to increase the chances of a cure and reduce the recurrence rate through photoimmunotherapy. © Fionn Große, private
Biologist Dr. Isis Wolf focuses on prostate cancer at the University Medical Centre Freiburg, aiming to increase the chances of a cure and reduce the recurrence rate through photoimmunotherapy. © Fionn Große, privateIt is a clever idea: a drug seeks out the tumour, binds to it and waits for a signal to destroy the tumour cells from within. The core of photoimmunotherapy (PIT) is a combination of biological precision with physical energy in the form of light.
This is exactly where Wolf’s research comes in. She uses a special protein – prostate-specific membrane antigen (PSMA) – as an anchor to ensure the therapy targets only cancer cells. PSMA is a protein that is found in extremely high density on the surface of prostate cancer cells, while healthy tissue produces very little.
It can also function as a biomarker to monitor disease progression. Dr. Wolf’s research group employs engineered antibodies that bind specifically to PSMA, like a key in a lock. "We have developed a photosensitiser – a fluorescent dye from the phthalocyanine family that is activated by light," Dr. Wolf explains. "By conjugating this dye to the antibodies, we create an antibody-drug conjugate, which is then directed specifically to cancer cells." This approach ensures that only tumour tissue is targeted. Additionally, the dye renders the targeted cells visible: under low-intensity excitation, they emit a red fluorescent signal when observed under a microscope.
Targeted cell death through photons
As long as no light falls on the conjugate, it remains completely inactive. The fluorescent dye is only activated when exposed to harmless red light with a wavelength of 692 nm, which leads to the destruction of the cell. At the molecular level, the light triggers a chain reaction: the activated dye damages the cancer cell so severely that pores form in its cell membrane. "Ions and water can enter through these pores, and after about five hours the cells eventually burst," says the biologist. "This type of cell death is called pyroptosis." The process is highly efficient not only because it acts extremely locally and spares surrounding tissue, but also because the tumour is simultaneously rendered visible by the fluorescent dye. In the future, this could help to remove the tumour even more precisely.
Wolf’s goal is to one day offer this therapy as an adjunct to radical prostatectomy in order to reduce the recurrence rate. Her idea is to administer the conjugate intravenously to the patient the day before surgery. Then, shortly after the prostate has been removed – and while it is being examined in pathology and the patient is still in the operating room – guide the surgical robot to the relevant area using a laser fibre to illuminate it. "This could be used as an add-on to target any remaining tumour cells," says Wolf, who was awarded the Klaus Mangold Prize for her work. Side effects would be minimal, as activation by the light would be limited to the surgical site.
Wake-up call for the immune system
What makes PIT a very promising approach is something known as immunogenic cell death. When cancer cells are destroyed by the light pulse, they release fragments of their contents along with danger signals. The immune system, which had previously ignored the cancer, suddenly recognises these signals as a threat, and immune cells are activated. It is as if the body were being 'vaccinated' from within.
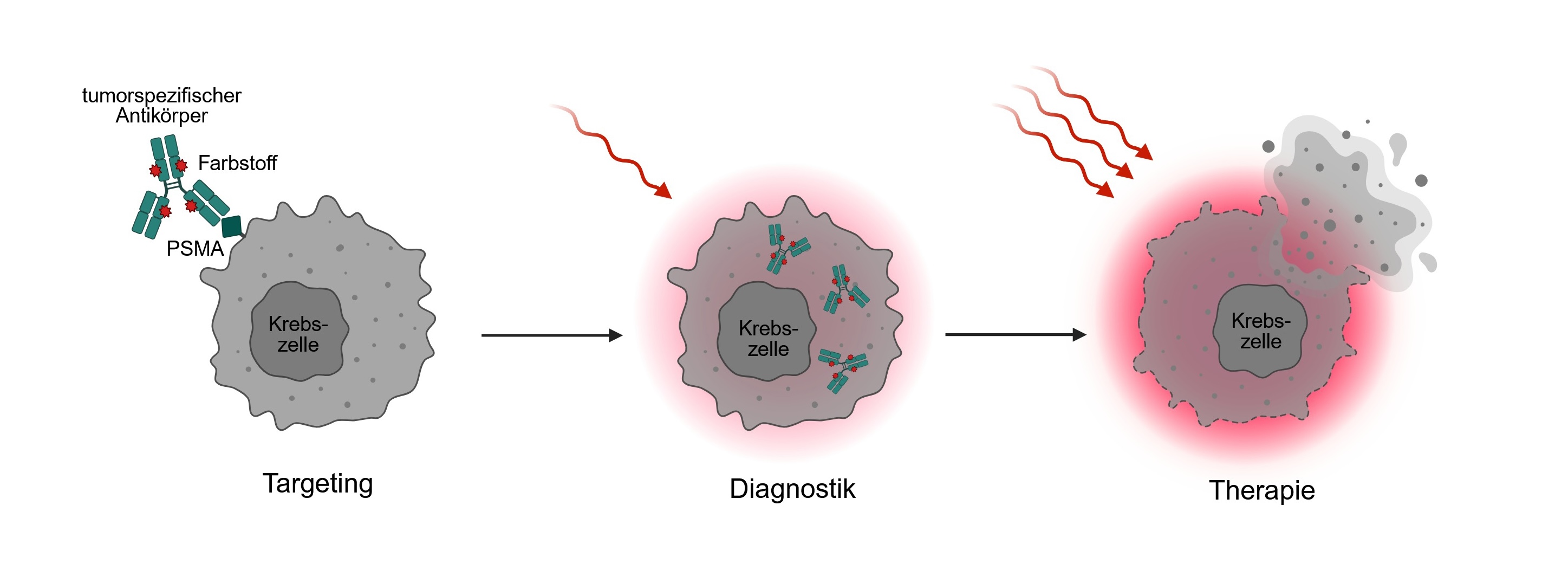 The conjugate of a tumour-specific antibody and a fluorescent dye binds to the PSMA structure on the tumour cell and is subsequently internalised. Upon exposure to infrared light, cell death (pyroptosis) is induced, resulting in cell lysis. © Created in BioRender. Wolf, I. T. (2026) https://BioRender.com/f1kvu6b
The conjugate of a tumour-specific antibody and a fluorescent dye binds to the PSMA structure on the tumour cell and is subsequently internalised. Upon exposure to infrared light, cell death (pyroptosis) is induced, resulting in cell lysis. © Created in BioRender. Wolf, I. T. (2026) https://BioRender.com/f1kvu6b
In the future, Wolf would like to use immunocompetent mice to investigate whether this immune system activation also has systemic effects. "I am hoping to be able to target hidden metastases that cannot be surgically removed," she explains. A potential side effect of immune stimulation, however, could be overstimulation, which would lead to a cytokine storm, as can occur with CAR-T cell therapy. Since PIT is intended to target only residual (remaining) tumour cells, Wolf considers this low risk. In a follow-up project, Wolf is planning to investigate exactly where the conjugate localises within the cell after uptake and what precisely happens to it.
Perspective beyond the prostate
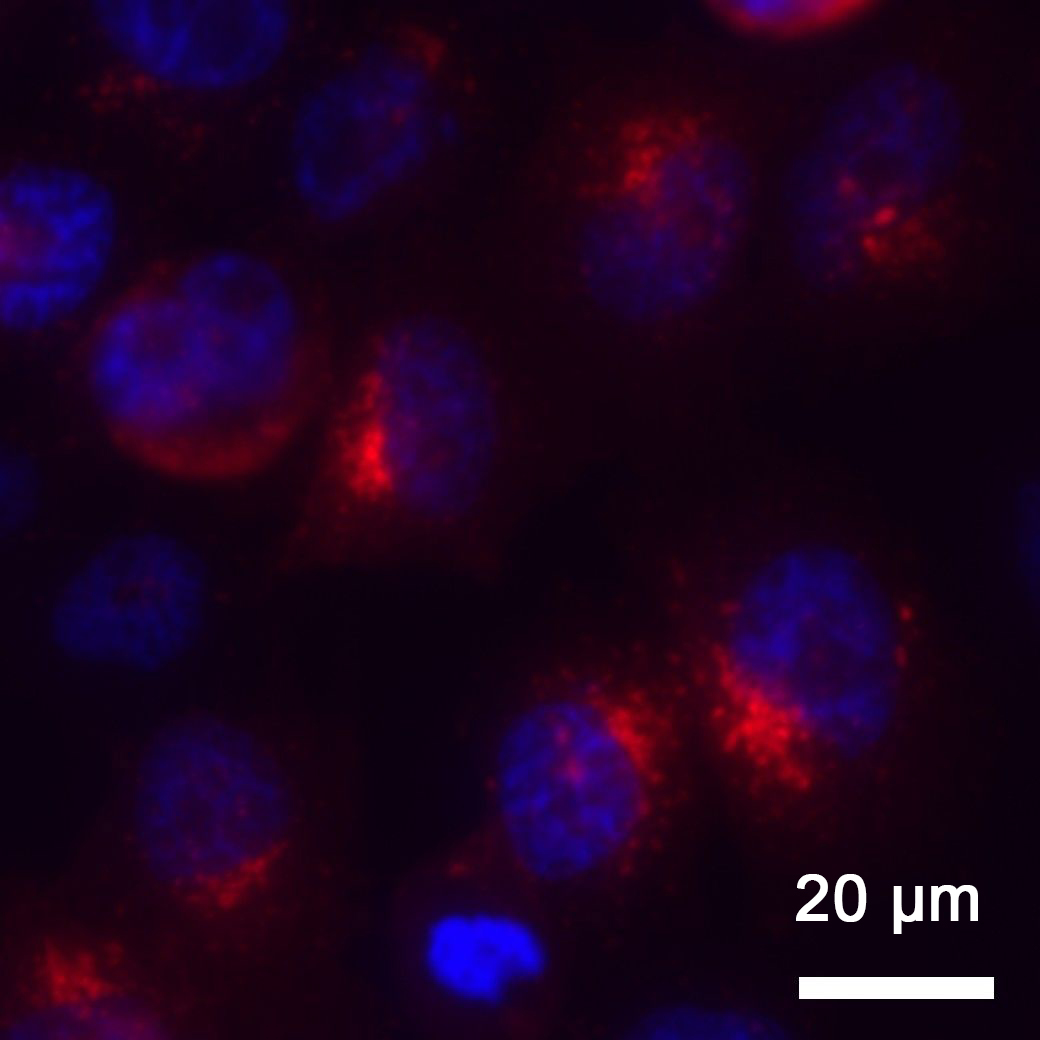 The tumour cells of the prostate carcinoma and their blue nuclei are visible under the microscope. The red fluorescence signals indicate the accumulation of the antibody-dye conjugate within the cells after it has bound to PSMA structures. © Dr. Isis Wolf, University Medical Centre Freiburg
The tumour cells of the prostate carcinoma and their blue nuclei are visible under the microscope. The red fluorescence signals indicate the accumulation of the antibody-dye conjugate within the cells after it has bound to PSMA structures. © Dr. Isis Wolf, University Medical Centre FreiburgDespite the positive results in the preclinical phase, the path to routine clinical application will be a long one. All components – antibody, dye and conjugate – must obtain individual clinical approval. Wolf hopes to build on already approved antibodies in order to reduce the development timeline. Assisted by a grant from the Scientific Society Freiburg, she is already testing the approach in colorectal cancer as well as targeting breast and bladder cancer. Photoimmunotherapy for prostate cancer could thus evolve into a modular system in which only the antibodies need to be exchanged to treat different tumour entities.
In December 2025, Wolf was awarded the Klaus Mangold Prize for her promising approach. Established in 2024 by the University Medical Centre Freiburg, the award recognises outstanding projects aimed at improving patient care. Wolf regards this prize as both recognition and motivation to further disseminate her research and continue working on an alternative to chemotherapy to make future cancer therapies more precise. Driven by the vision of a gentler cure, she talks about her future goal: "My hope is that our construct may one day be used in clinical practice, reduce recurrence rates and make treatment less of a burden for patients."