Detecting drug resistance of tumour cells
AI-assisted diagnostics declares war on lung cancer
Lung cancer is one of the most common cancers in Germany and has a particularly high mortality rate. A significant challenge in treating this disease lies in the resistance of lung tumours to conventional drug therapies, rendering chemotherapy ineffective. However, there is hope on the horizon as a team of experts from Baden-Württemberg, consisting of hospital representatives, researchers and industry partners, has joined forces under the umbrella of a project funded by the German Federal Ministry for Economic Affairs and Climate Action. Their objective is to develop an innovative AI-supported test procedure that paves the way for individualised therapy approaches for lung cancer patients.
According to the Robert Koch Institute, 35,300 men and 21,900 women were diagnosed with lung cancer in Germany alone in 2018 (number of new cases).1) Five years after initial diagnosis, only around 22 percent of the women and 17 percent of the men were still alive. This clearly demonstrates a significant need for new diagnostic approaches that address the unique challenges of lung cancer. "The most common type of lung cancer is called non-small cell lung carcinoma (NSCLC), which is the leading cause of death among cancer patients. At the same time, NSCLC exhibits a high tendency for tumour cell-intrinsic drug resistance. The limited efficacy of conventional chemotherapy in this context is a significant challenge," explains Dr. Martin Kriebel, a molecular and neurobiologist who is a project leader at the NMI Natural and Medical Sciences Institute in Reutlingen. Alongside a team led by Sergey Biniaminov, CEO at HS Analysis, an AI company from Karlsruhe, and the Schwarzwald-Baar Klinikum in Villingen-Schwenningen, Kriebel is actively developing an innovative AI-based test procedure as part of the IDOL (AI-based diagnostics of lung cancer to support personalised therapy decisions) funding project, which aims to predict drug resistances.
 The aim of the innovative AI-supported test system is to assist doctors in the future in choosing the most effective treatment approach for each patient’s unique circumstances. © Schwarzwald-Baar Klinikum
The aim of the innovative AI-supported test system is to assist doctors in the future in choosing the most effective treatment approach for each patient’s unique circumstances. © Schwarzwald-Baar KlinikumCurrently, there are no traditional laboratory tests for detecting drug resistance in lung cancer cells. "The project aims to address this gap by developing a new AI-supported solution, which will subsequently be marketed by HS Analysis GmbH," Biniaminov explains. As chemotherapy can be physically demanding and challenging for patients, it is crucial to predict the likelihood of therapy success or whether alternative treatment options should be considered. "The IDOL project strives to come up with a reliable decision-making tool for doctors," Kriebel adds.
The project is receiving funding of 400,000 euros over two years through the ZIM programme (Central Innovation Programme for SMEs) run by the German Federal Ministry for Economic Affairs and Climate Action. "Collaborative efforts within the BioLAGO e.V. health network played a vital role in refining the initial project idea. BioLAGO put us in contact with the NMI and the Schwarzwald-Baar Klinikum. This step was crucial in enabling the necessary testing of the test procedure on relevant tissue samples obtained from patients," Biniaminov continues.
First project successes: positive in vitro data available
The project got underway in 2021, and the Schwarzwald-Baar Klinikum is responsible for recruiting and classifying affected patients. The clinic is also providing sample materials from tumour tissues that can be classified as NSCLC. The NMI will use these samples to conduct tests using a combined histological method to detect drug resistance in lung cancer.
The method involves detecting the activity of a specific molecular signalling pathway known to be highly active in NSCLC, and which leads to drug resistance and impedes the effectiveness of chemotherapy. Using diagnostic biomarkers, the NMI is developing a sensitive test system for detecting the activation status of this signalling pathway. Multiple parameters of the signalling pathway activity are taken into consideration to ensure the accuracy and reliability of the test system.
"In the first year of the IDOL project, at the NMI we have already successfully established the desired detection method using model cell lines. This has already yielded the first positive in vitro data," said Kriebel. These in vitro data serve as initial evidence that the test method functions and thus pave the way for applying the detection method on relevant histological sample materials from clinical settings.
AI system supports informed therapy decisions
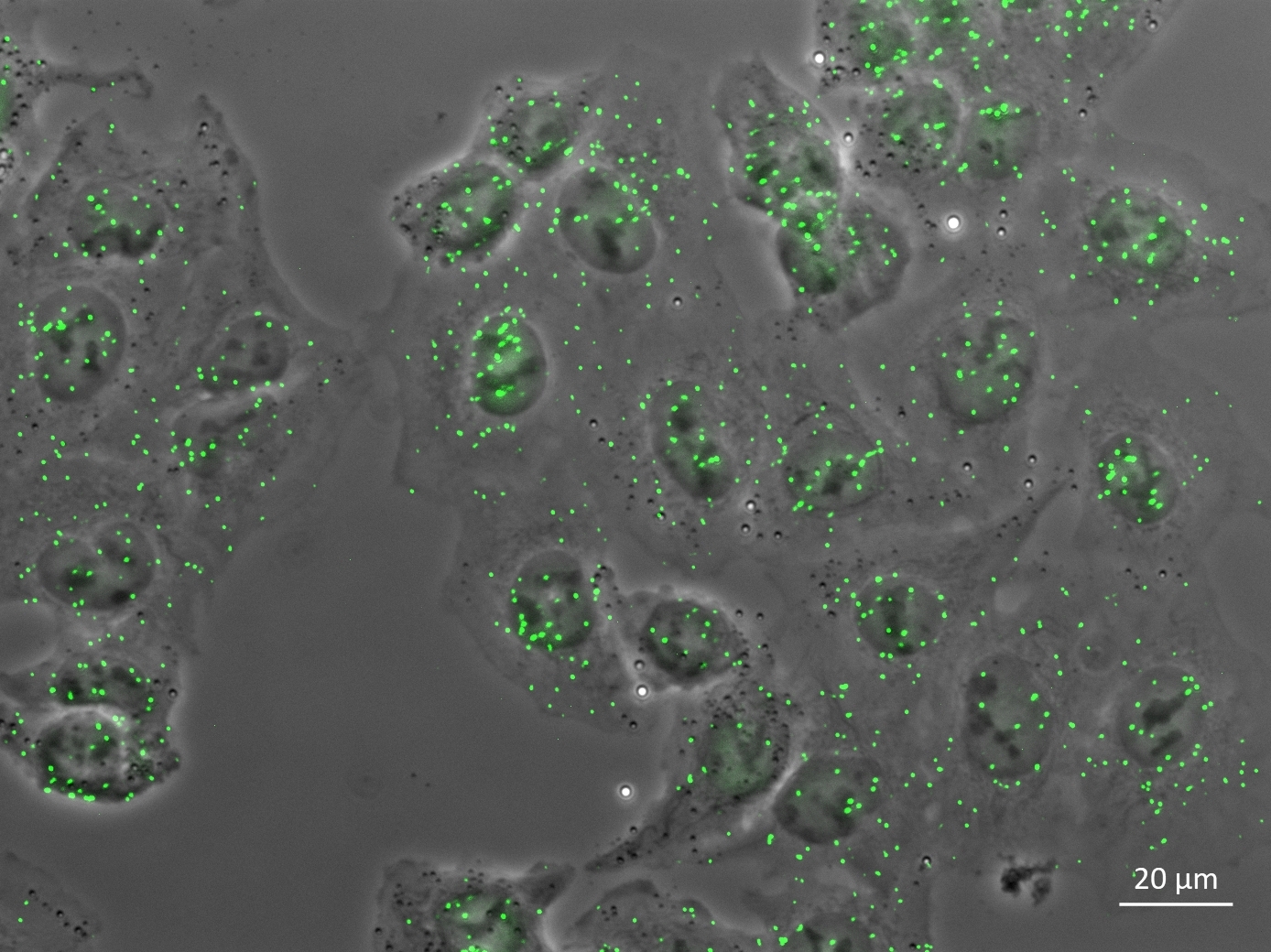 The detection method visualises the activity status of signalling molecules © NMI
The detection method visualises the activity status of signalling molecules © NMIIn the subsequent phase, the histological detection method developed at the NMI will be integrated into an automated AI-supported quantitative image analysis by the experts at HS Analysis. The ultimate objective is to create an innovative image- and AI-based method that is capable of quantifying complex biomedical processes at the cellular level. The AI system analyses microscopic images of cells that have been stained to highlight the active signalling pathways. These visual data are supplemented with additional medical parameters related to lung tumours, which are then quantified and processed numerically. These data form a crucial foundation for the AI’s deep learning capabilities, enabling large and complex amounts of data to be collected and comprehensively evaluated. This makes the detection procedure even more robust and reliable over time, providing doctors with valuable information to make informed therapy decisions.
From prototype to finished test system
"The aim is to develop a complete test system comprising reagents and analysis software that can be easily integrated into existing laboratory process," Biniaminov explains. The project partners attach great importance to being able to minimise any additional effort and streamline procedures. The test system will utilise sample material obtained from routine tissue biopsies that have undergone histological processing in routine standard laboratory diagnostics. It is not necessary to collect new sample material specifically for this test.
"Our overarching goal is to make the innovative test system available to as many lung cancer patients as possible. But there is still a considerable journey ahead," concludes Kriebel. By the end of the funding phase in 2024, the project partners are aiming to have developed a first prototype providing tangible evidence of the undeniable effectiveness of the new AI-based method. The next critical milestone will then be the transition to market entry, which will be undertaken by HS Analysis GmbH. This will pave the way for widespread adoption of the innovative test system.